Abstract
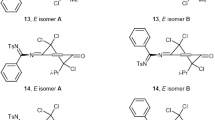
Reactions of halogens with N-arylsulfonyl-1,4-benzoquinone monoimines occur with the formation of a halogenonium ion that either transforms into a carbocation where the first halogen atom adds to the carbon in the ortho-position relative to the carbonyl carbon, or the halogenonium ion adds directly the second halogen atom.
Similar content being viewed by others
References
Avdeenko, A.P. and Konovalova, S.A., Zh. Org. Khim., 2006, vol. 42, p. 669
Avdeenko, A.P. and Konovalova, S.A., Il’chenko, A.Ya., and Glinyanaya, N.M., Zh. Org. Khim., 2006, vol. 42, p. 64.
Avdeenko, A.P. and Konovalova, S.A., Zh. Org. Khim., 2006, vol. 42, p. 364.
Burmistrov, S.I. and Titov, E.A., Zh. Obshch. Khim., 1952, vol. 22, p. 999.
Adams, R. and Reifschneider, W., Bull. Soc. Chim., 1958, p. 23; Adams, R. and Blomstrom, D., J. Am. Chem. Soc., 1953, vol. 75, p. 3405.
Author information
Authors and Affiliations
Additional information
Original Russian Text © A.P. Avdeenko, S.A. Konovalova, O.N. Ludchenko, 2006, published in Zhurnal Organicheskoi Khimii, 2006, Vol. 42, No. 5, pp. 702–707.
For communication V, see [1].
Rights and permissions
About this article
Cite this article
Avdeenko, A.P., Konovalova, S.A. & Ludchenko, O.N. Halogenation of N-substituted para-quinone monoimine and para-quinone monooxime esters: VI. Regular trends in chlorination and bromination of N-arylsulfonyl-1,4-benzoquinone monoimines alkyl-substituted in the quinoid ring. Russ J Org Chem 42, 683–688 (2006). https://doi.org/10.1134/S107042800605006X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S107042800605006X