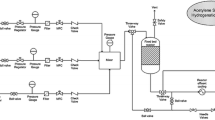
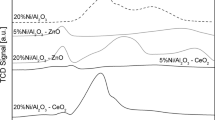
Abstract
The effect that calcination of an alumina–chromia catalyst containing 13 wt % Cr with additions of Na+ and Zr4+ in an air–water vapor atmosphere (from 0 to 80 vol % water vapor) at 750°С and a pressure of 1 bar exerts on the physicochemical properties of the catalyst and its activity in n-butane dehydrogenation was studied. The steam treatment leads to a slight decrease in the specific surface area (by up to 10%), partial decomposition of Cr(VI) compounds (up to 60%), and Cr2O3 crystallization. The catalytic activity decreases with an increase in the water vapor : air ratio. Low water vapor concentration (10 vol %) favors a significant decrease in the amount of the coke formed (by 60%) without significantly affecting the yield of alkenes. Thus, introduction of water vapor into the calcination atmosphere allows control of the Cr(VI) amount and catalyst selectivity.




Similar content being viewed by others
REFERENCES
Sattler, J.J.H.B., Ruiz-Martinez, J., Santillan-Jimenez, E., and Weckhuysen, B.M., Chem. Rev., 2014, vol. 114, no. 20, pp. 10613–10653. https://doi.org/10.1021/cr5002436
Sanfilippo, D., Catal. Today, 2011, vol. 178, no. 1, pp. 142–150. https://doi.org/10.1016/j.cattod.2011.07.013
Busca, G., Adv. Catal., 2014, vol. 57, pp. 319–404. https://doi.org/10.1016/B978-0-12-800127-1.00003-5
Nazimov, D.A., Klimov, O.V., Danilova, I.G., Trukhan, S.N., Saiko, A.V., Cherepanova, S.V., Chesalov, Y.A., Martyanov, O.N., and Noskov, A.S., J. Catal., 2020, vol. 391, pp. 35–47. https://doi.org/10.1016/j.jcat.2020.08.006
Gorriz, O.F., Cortes Corberan, V., and Fierro, J.L.G., Ind. Eng. Chem. Res., 1992, vol. 31, no. 12, pp. 2670–2674. https://doi.org/10.1021/ie00012a007
Rodemerck, U., Kondratenko, E.V., Otroshchenko, T., and Linke, D., Chem. Commun., 2016, vol. 52, no. 82, pp. 12222–12225. https://doi.org/10.1039/C6CC06442F
Rombi, E., Cutrufello, M.G., Solinas, V., Rossi, S. De, Ferraris, G., and Pistone, A., Appl. Catal. A, 2003, vol. 251, no. 2, pp. 255–266. https://doi.org/10.1016/S0926-860X(03)00308-9
Cavani, F., Koutyrev, M., Trifirò, F., Bartolini, A., Ghisletti, D., Iezzi, R., Santucci, A., and Del Piero, G., J. Catal., 1996, vol. 158, no. 1, pp. 236–250. https://doi.org/10.1006/jcat.1996.0023
Nazimov, D.A., Klimov, O.V., Saiko, A.V., Trukhan, S.N., Glazneva, T.S., Prosvirin, I.P., Cherepanova, S.V., and Noskov, A.S., Catal. Today, 2021, vol. 375, pp. 401–409. https://doi.org/10.1016/j.cattod.2020.03.005
Bocanegra, S.A., Castro, A.A., Guerrero-Ruíz, A., Scelza, O.A., and De Miguel, S.R., Chem. Eng. J., 2006, vol. 118, no. 3, pp. 161–166. https://doi.org/10.1016/j.cej.2006.02.004
Wang, G., Song, N., Lu, K., Wang, W., Bing, L., Zhang, Q., Fu, H., Wang, F., and Han, D., Catalysts, 2019, vol. 9, no. 11, p. 968. https://doi.org/10.3390/catal9110968
Masson, J., Bonnier, J.M., Duvigneaud, P.H., and Delmon, B., J. Chem. Soc., Faraday Trans. 1, 1977, vol. 73, pp. 1471–1479. https://doi.org/10.1039/F19777301471
Rombi, E., Gazzoli, D., Cutrufello, M.G., De Rossi, S., and Ferino, I., Appl. Surf. Sci., 2010, vol. 256, no. 17, pp. 5576–5580. https://doi.org/10.1016/j.apsusc.2009.12.151
Patent US 2399678A, Publ. 1946.
Patent US 2419997A, Publ. 1947.
Jóźwiak, W.K. and Dalla Lana, I.G., J. Chem. Soc., Faraday Trans., 1997, vol. 93, no. 15, pp. 2583–2589. https://doi.org/10.1039/a608563f
McDaniel, M.P., J. Catal., 1982, vol. 76, no. 1, pp. 37–47. https://doi.org/10.1016/0021-9517(82)90234-2
Bartholomew, C.H., Appl. Catal. A, 2001, vol. 212, nos. 1–2, pp. 17–60. https://doi.org/10.1016/S0926-860X(00)00843-7
Anderson, P.J. and Morgan, P.L., Trans. Faraday Soc., 1964, vol. 60, pp. 930–937. https://doi.org/10.1039/TF9646000930
Arai, H. and Machida, M., Appl. Catal. A, 1996, vol. 138, no. 2, pp. 161–176. https://doi.org/10.1016/0926-860X(95)00294-4
Fridman, V.Z., Xing, R., and Severance, M., Appl. Catal. A, 2016, vol. 523, pp. 39–53. https://doi.org/10.1016/j.apcata.2016.05.008
Patent US 8680357B1, Publ. 2014.
Airaksinen, S.M.K., Krause, A.O.I., Sainio, J., Lahtinen, J., Chao, K.J., Guerrero-Pérez, M.O., and Bañares, M.A., Phys. Chem. Chem. Phys., 2003, vol. 5, no. 20, pp. 4371–4377. https://doi.org/10.1039/B305802F
Weckhuysen, B.M., Schoonheydt, R.A., Jehng, J.-M., Wachs, I.E., Cho, S.J., Ryoo, R., Kijlstra, S., and Poels, E., J. Chem. Soc., Faraday Trans., 1995, vol. 91, no. 18, pp. 3245–3253. https://doi.org/10.1039/FT9959103245
Kanervo, J.M. and Krause, A.O.I., J. Catal., 2002, vol. 207, no. 1, pp. 57–65. https://doi.org/10.1006/jcat.2002.3531
Weckhuysen, B.M., Ridder, L.M. De, and Schoonheydt, R.A., J. Phys. Chem., 1993, vol. 97, no. 18, pp. 4756–4763. https://doi.org/10.1021/j100120a030
Egorova, S.R., Bekmukhamedov, G.E., and Lamberov, A.A., Kinet. Catal., 2013, vol. 54, no. 1, pp. 49–58. https://doi.org/10.1134/S0023158413010072
Weckhuysen, B.M. and Schoonheydt, R.A., Catal. Today, 1999, vol. 51, no. 2, pp. 223–232. https://doi.org/10.1016/S0920-5861(99)00047-4
Puurunen, R.L. and Weckhuysen, B.M., J. Catal., 2002, vol. 210, no. 2, pp. 418–430. https://doi.org/10.1006/jcat.2002.3686
Fridman, V.Z. and Xing, R., Appl. Catal. A, 2017, vol. 530, pp. 154–165. https://doi.org/10.1016/j.apcata.2016.11.024
Fridman, V.Z. and Xing, R., Ind. Eng. Chem. Res., 2017, vol. 56, no. 28, pp. 7937–7947. https://doi.org/10.1021/acs.iecr.7b01638
Hakuli, A., Kytökivi, A., Krause, A.O.I., and Suntola, T., J. Catal., 1996, vol. 161, no. 1, pp. 393–400. https://doi.org/10.1006/jcat.1996.0197
De Rossi, S., Ferraris, G., Fremiotti, S., Cimino, A., and Indovina, V., Appl. Catal. A, 1992, vol. 81, no. 1, pp. 113–132. https://doi.org/10.1016/0926-860X(92)80264-D
De Rossi, S., Ferraris, G., Fremiotti, S., Garrone, E., Ghiotti, G., Campa, M.C., and Indovina, V., J. Catal., 1994, vol. 148, no. 1, pp. 36–46. https://doi.org/10.1006/jcat.1994.1183
Hakuli, A., Kytökivi, A., and Krause, A.O.I., Appl. Catal. A, 2000, vol. 190, nos. 1–2, pp. 219–232. https://doi.org/10.1016/S0926-860X(99)00310-5
Weckhuysen, B.M., Ridder, L.M. De, Grobet, P.J., and Schoonheydt, R.A., J. Phys. Chem., 1995, vol. 99, no. 1, pp. 320–326. https://doi.org/10.1021/j100001a048
Matsunaga, Y., Bull. Chem. Soc. Jpn., 1957, vol. 30, no. 8, pp. 868–872. https://doi.org/10.1246/bcsj.30.868
Dumez, F.J. and Froment, G.F., Ind. Eng. Chem. Process Des. Dev., 1976, vol. 15, no. 2, pp. 291–301. https://doi.org/10.1021/i260058a014
Airaksinen, S.M.K., Bañares, M.A., and Krause, A.O.I., J. Catal., 2005, vol. 230, no. 2, pp. 507–513. https://doi.org/10.1016/j.jcat.2005.01.005
ACKNOWLEDGMENTS
The authors are grateful to Cand. Sci. (Phys.-Math.) S.V. Cherepanova, Cand. Sci. (Phys.-Math.) T.V. Larina, and T.Ya. Efimenko for participation in studying the samples.
Funding
The study was performed within the framework of the government assignment for the Institute of Catalysis, Siberian Branch, Russian Academy of Sciences (project АААА-А21-121011890074-4).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated from Zhurnal Prikladnoi Khimii, No. 9, pp. 1195–1203, January, 2021 https://doi.org/10.31857/S0044461821090097
Rights and permissions
About this article
Cite this article
Nazimov, D.A., Klimov, O.V., Saiko, A.V. et al. Effect of Steam–Air Treatment of Alumina–Chromia Dehydrogenation Catalysts on Their Physicochemical and Catalytic Characteristics. Russ J Appl Chem 94, 1283–1291 (2021). https://doi.org/10.1134/S1070427221090111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427221090111