Abstract
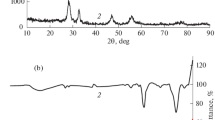
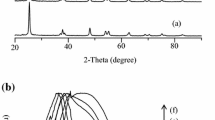
This study evaluated a removal efficiency of NLFX from aqueous solutions using a photocatalytic process. In this study, Bi2WO6 were synthesized by microwave method and Ce was loaded on Bi2WO6 by wet inoculation method. The prepared photocatalysis were characterized by XRD, FTIR, SEM, EDS/Map, TEM, BET/BJH, DRS-UV/Vis analysis. The degradation of NFLX was investigated by the photocatalysis synthesized under solar light. Among the photocatalysis, Ce/Bi2WO6 (8%) had the highest NFLX degradation efficiency. Response surface methodology (RSM) with central composite design (CCD) was used to investigate pH, time, mass of catalyst Ce/Bi2WO6 (8%) g, and NFLX concentration. In optimal conditions (pH 3.75, time 126.11 min, mass of Ce/Bi2WO6 (8%) 0.03 g, and NFLX concentration 10.11 mg/L) the photocatalytic degradation percentages of NFLX were found to be 99.62%. Isothermic studies have shown that Fritz–Schlunder isotherms have the best fit with Ce/Bi2WO6 (8%) data (the least error).














Similar content being viewed by others
Change history
05 October 2021
An Erratum to this paper has been published: https://doi.org/10.1134/S107042722107020X
REFERENCES
Payan, A., Isari, A.A., and Gholizade, N., Chem. Eng. J., 2019, vol. 361, p. 1121.
Wan, Z., Hu, J., and Wang, J., J. Environ. Manage., 2016, vol. 182, p. 284.
Farzadkia, M., Rahmani, K., Gholami, M., Esrafili, A., Rahmani, A., and Rahmani, H., Korean J. Chem. Eng., 2014, vol. 31, p. 2014.
Daghrir, R. and Drogui, P., Environ., Chem. Letter., 2013, vol. 3, p. 209.
Sudhaik, A., Raizada, P., Shandilya, P., and Singh, P., J. Environ. Chem. Eng., 2018, vol. 6, p. 3874.
Caro, E., Marce, R.M., Cormack, P.A., Sherrington, D.C., and Borrull, F., Anal. Chim. Acta, 2005, vol. 3, p. 81.
Chen, X. and Wang, J., Chem. Eng. J., 2020, vol. 395, p. 125095.
Li, Y., Liu, J., Huang, X., and Li, G., Cryst. Growth Des., 2007, vol. 7, p. 1350.
Zhang, C. and Zhu, Y., Chem. Mater., 2005, vol. 17, p. 3537.
Shang, M., Wang, W., Sun, S., Zhou, L., and Zhang, L., J. Physical Chemistry, C, vol. 17, 10407, p. 2008.
Fu, H., Zhang, L., Yao, W., and Zhu, Y., Appl. Catal. B: Environ., 2006, vol. 66, p. 10.
Yu, J., Xiong, J., Cheng, B., Yu, Y., and Wang, J., J. Solid State Chem., 2005, vol. 178, p. 1968.
Fard, N.E., Fazaeli., R., Yousefi, M., and Abdolmoham-madi, S., Russ. J. Physic. Chem., A, 2021, vol. 95, p. 23.
Huang, H., Liu, K., Chen, K., Zhang, Y., Zhang, Y., and Wang, S., J. Phys. Chem., 2014, vol. 118, 2014, p. 14379.
Kashi, N., Fard, N.E., and Fazaeli, R., Russ. J. Appl. Chem., 2017, vol. 90, p. 977.
Fard, N.E. and Fazaeli, R., Iran. J. Catal., 2018, vol. 8, p. 133.
Fard, N.E. and Fazaeli, R., Russ. J. Physic. Chem., A, 2018, vol. 92, p. 2835.
Fazaeli, R. and Fard, N.E., Russ. J. Appl. Chem., 2020, vol. 93, p. 973.
Fard, N.E., Fazaeli, R., and Ghiasi, R., Chem. Eng. Technol., 2016, vol. 39, p. 149.
Saadi, R., Saadi, Z., Fazaeli, R., and Fard, N.E., Korean J. Chem. Eng., 2015, vol. 32, p. 787.
Dolgonos, A., Mason, T.O., and Poeppelmeier, K.R., J. Solid State Chem., 2016, vol. 240, p. 43.
Viezbicke, B.D., Patel, S., Davis, B.E., and Birnie, D.P., Phys. Status Solidi B, 2015, vol. 252, p. 1700.
Fard, N.E., Fazaeli., R., Yousefi, M., Abdolmohammadi, S., Appl. Physic., A, 2019, vol. 125, p. 632.
Fard, N.E., Fazaeli., R., Yousefi, M., Abdolmohammadi, S., Chem. Select., 2019, vol. 4, p. 9529.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest requiring disclosure in this article.
Rights and permissions
About this article
Cite this article
Fekra, S.S., Fard, N.E. & Fazaeli, R. Photocatalytic Degradation of Antibiotic Norfloxacin Aqueous Solution by Ce/Bi2WO6: Optimization and Simulation of Process by RSM. Russ J Appl Chem 94, 824–834 (2021). https://doi.org/10.1134/S1070427221060161
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427221060161