Abstract
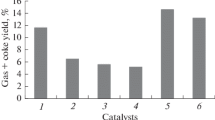
Effect of mixed Mg–Al oxides contained in catalysts on the distribution of target products formed in cracking of a mixture of vacuum gas oil with sunflower oil was examined. It was found that the process of deoxygenation of oxygen-containing compounds can be controlled by varying the molar ratio between magnesium and aluminum in the mixed oxides. For catalyst samples containing mixed oxides with large Mg: Al molar ratios, the content of carbon dioxide in inorganic products grows, which is indicative of the increased activity of the decarboxylation reaction. In addition, the results of catalytic tests demonstrate that the highest conversion and yield of the benzine fraction are observed for catalyst samples containing mixed oxides with small magnesium-to-aluminum ratios of 0.32: 1 and 0.49: 1. A high activity of hydrogen-transfer reactions is observed for these samples, which leads to a decrease in the yield of light olefins. Thus, it is advisable, for obtaining an increased yield of C3 and C4 olefins, to include into the composition of the catalysts mixed oxides with increased content of magnesium, with Mg: Al ratios of 1.90: 1 and more.
Similar content being viewed by others
References
Doronin, V.P., Sorokina, T.P., Potapenko, O.V., Lipin, P.V., Dmitriev, K.I., Korotkova, N.V., and Gur’evskikh, S.Yu., Kataliz Prom-sti, 2016, vol. 16, no. 6, pp. 71–76. https://doi.org/10.18412/1816-0387-2016-6-71-76
Bobkova, T.V., Potapenko, O.V., Sorokina, T.P., and Doronin, V.P., Russ. J. Appl. Chem., 2017, vol. 90, no. 12, pp. 1900–1907. https://doi.org/10.1134/S1070427217120023.
Doronin, V.P., Lipin, P.V., Potapenko, O.V., Vysotskii, V.V., Gulyaeva, T.I., and Sorokina, T.P., Catal. Ind., 2018., vol. 10, no. 4, pp. 335–343. https://doi.org/10.1134/S2070050418040050.
Scherzer, J., Appl. Catal., 1991, vol. 75, no. 1, pp. 1–32. https://doi.org/10.1016/S0166-9834(00)83119-X
Corma, A., Palomares, A.E., Rey, F., and Márquez, F., J. Catal., 1997, vol. 170, no. 1, pp. 140–149. https://doi.org/10.1006/jcat.1997.1750
Palomares, A.E., López-Nieto, J.M., Lázaro, F.J., López, A., and Corma, A., Appl. Catal., B, 1999, vol. 20, no. 4, pp. 257–266. https://doi.org/10.1016/S0926-3373(98)00121-0
Climent, M.J., Corma, A., Iborra, S., Epping, K., and Velty, A., J. Catal., 2004, vol. 225, no. 2, pp. 316–326. https://doi.org/10.1016/j.jcat.2004.04.027
Polato, C.M.S., Henriques, C.A., Rodrigues, A.C.C., and Monteiro, J.L.F., Catal. Today, 2008, vol. 133–135, pp. 534–550. https://doi.org/10.1016/j.cattod.2007.12.046
Wang, D., Zhang, X., Cong, X., Liu, S., and Zhou, D., Appl. Catal., A, 2018, vol. 555, pp. 36–46. https://doi.org/10.1016/j.apcata.2018.02.009
Kostov, I., Mineralogiya (Metallurgy), Moscow: Mir, 1971, pp. 208–268.
Cavani, F., Trifiro, F., and Vaccari, A., Catal. Today, 1991, vol. 11, no. 2, pp. 173–301. https://doi.org/10.1016/0920-5861(91)80068-K
Layered Double Hydroxides, Duan, X. and Evans, D.G., Eds., New York: Springer, 2006. Struct. Bonding, vol. 119, pp. 1–87.
Saber, O. and Tagaya, H., Rev. Adv. Mater. Sci., 2005, vol. 10, no. 1, pp. 59–63.
Melero, J.A., Clavero, M.M., Calleja, G., García, A., Miravalles, R., and Galindo, T., Energy Fuels, 2010, vol. 24, pp. 707–717. https://doi.org/10.1021/ef900914e
Bielansky, P., Weinert, A., Schönberger, C., and Reichhold, A., Fuel Process. Technol., 2011, vol. 92, no. 12, pp. 2305–2311. https://doi.org/10.1016/j.fuproc.2011.07.021
Doronin, V.P., Potapenko, O.V., Lipin, P.V., and Sorokina, T.P., Fuel, 2013, vol. 106, pp. 757–765. https://doi.org/10.1016/j.fuel.2012.11.027
Lovás, P., Hudec, P., Hadvinová, M., and Ház, A., Fuel Process. Technol., 2015, vol. 134, pp. 223–230. https://doi.org/10.1016/j.fuproc.2015.01.038
Abbasov, V., Mammadova, T., Aliyeva, N., Abbasov, M., Movsumov, N., Joshi, A., Lvov, Y., and Abdullayev, E., Fuel, 2016, vol. 181, pp. 55–63. https://doi.org/10.1016/j.fuel.2016.04.088
Doronin, V.P., Lipin, P.V., Potapenko, O.V., Zhuravlev, Ya.E., and Sorokina, T.P., Khim. Interesakh Ustoich. Razvit., 2017, no. 25, pp. 385–392. https://doi.org/10.15372/khur20170404
Dupain, X., Costa, D.J., Schaverien, C.J., Makkee, M., and Moulijn, J.A., Appl. Catal., B, 2007, vol. 72, nos. 1–2, pp. 44–61. https://doi.org/10.1016/j.apcatb.2006.10.005
Na, J.-G., Yi, B.E., Kim, J.N., Yi, K.B., Park, S.-Y., Park, J.-H., Kim, J.-N., and Ko, C.H., Catal. Today, 2010, vol. 156, nos. 1–2, pp. 44–48. https://doi.org/10.1016/j.cattod.2009.11.008
RF Patent 2 300 420 (publ. 2007).
Potapenko, O.V., Doronin, V.P., Sorokina, T.P., Talsi, V.P., and Likholobov, V.A., Appl. Catal., B, 2012, vols. 117–118, pp. 177–184. https://doi.org/10.1016/j.apcatb.2012.01.014
Belaya, L.A., Doronin, V.P., Sorokina, T.P., and Gulyaeva, T.I., Russ. J. Appl. Chem., 2009, vol. 82, no. 2, pp. 236–242. https://doi.org/10.1134/S1070427209020141
Basila, M.R. and Kantner, T.R., J. Phys. Chem., 1966, vol. 70, no. 5, pp. 1681–1682. https://doi.org/10.1021/j100877a514
Doronin, V.P. and Sorokina, T.P., Ros. Khim. Zh., 2007, vol. LI, no. 4, pp. 23–28.
Catal. Today, 1989, vol. 5, no. 1, pp. 1–120. https://doi.org/10.1016/0920-5861(89)80034-3
Katikaneni, S.P.R., Adjaye, J.D., Idem, R.O., and Bakhshi, N.N., Ind. Eng. Chem. Res., 1996, vol. 35, no. 10, pp. 3332–3346. https://doi.org/10.1021/ie950740u
Maher, K.D. and Bressler, D.C., Bioresour. Technol., 2007, vol. 98, no. 12, pp. 2351–2368. https://doi.org/10.1016/j.biortech.2006.10.025
Immer, J.G., Kelly, M.J., and Lamb, H.H., Appl. Catal., A, 2010, vol. 375, no. 1, pp. 134–139. https://doi.org/10.1016/j.apcata.2009.12.028
Bernas, H., Eränen, K., Simakova, I., Leino, A.-R., Kordas, K., Myllyoja, J., Maki-Arvela, P., Salmi, T., and Murzin, D.Yu., Fuel, 2010, vol. 89, no. 8, pp. 2033–2039. https://doi.org/10.1016/j.fuel.2009.11.006
Renz, M., Eur. J. Org. Chem., 2005, vol. 2005, no. 6, pp. 979–988. https://doi.org/10.1002/ejoc.200400546
Funding
The study was carried out und the State assignment of the Institute of Hydrocarbon Processing, Siberian Division, Russian Academy of Sciences inconformity with the Program of Basic Research of State Academies of Sciences for the years of 2013–2020 in the direction V.47, project no. V.47.1.3 [State registration number in the EGISU NIOKTR (Unified State Information System for Accounting R&D and technological activities) AAAA-A17-117021450099-9].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors state that they have no conflict of interest to be disclosed in the present communication.
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Prikladnoi Khimii, 2019, Vol. 92, No. 10, pp. 1291–1300.
Rights and permissions
About this article
Cite this article
Lipin, P.V., Potapenko, O.V., Sorokina, T.P. et al. Catalytic Cracking of a Mixture of Vacuum Gas Oil and Vegetable Oil in the Presence of Mixed Mg–Al Oxide of Various Compositions. Russ J Appl Chem 92, 1383–1391 (2019). https://doi.org/10.1134/S1070427219100082
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427219100082