Abstract
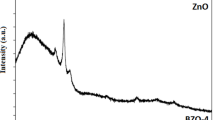
Boron doped vanadium thin films were fabricated on the micro-slide glass substrates by spray pyrolysis technique at substrate temperature of 400°C. Initially, 0.1 M vanadium(III) chloride (VCl3) solution was prepared in ethanol: water mixture (1: 4) solvent. To prepare the boron doped vanadium oxide films as the concentrations of 2, 5, 7, 10, 20%, the suitable amount of H3BO3 was added in 0.1 M VCl3 solution for each of the samples. X-Ray diffraction experiment with the produced films showed that tetragonal β-V2O5 phases formed. The synthesized boron-doped V2O5 thin films having the large surface area demonstrated the efficient catalytic properties in the photocatalytic degradation of methyl blue in water samples under Xenon light. The photocatalytic reaction efficiency was measured by recording the decrease of absorbance at 590 nm in UV-Vis absorption spectra. The methyl blue dye was degradated in approximately 30 min. The photocatalytic experiment results of the produced thin films showed that boron doping amount positively effects the degradation efficiency and reaction time.
Similar content being viewed by others
References
Eskalen, H., Kursun, C., Asian, M., Cesme, M., and Gogebakan, M., arXiv Preprint: 170906941, 2017.
Chong, M.N., Jin, B., Chow, C.W., and Saint, C., Water Res., 2010, vol. 44, pp. 2997–3027.
Xiang, Q., Lang, D., Shen, T., and Liu, F., Applied Catalysis B: Environmental, 2015, vol. 162, pp. 196–203.
Konstantinou, I.K. and Albanis, T.A., Applied Catalysis B: Environmental, 2004, vol. 49, pp. 1–14.
Yagub, M.T., Sen, T.K., Afroze, S., and Ang, H.M., Advances in Colloid and Interface Science, 2014, vol. 209, pp. 172–184.
Isleyen, M., Ilkme, E.S., and Soylu, G.S.P., Korean J. Chem. Eng., 2017, vol. 34, pp. 1786–1792.
Behpour, M., Mehrzad, M., and Hosseinpour-Mashkani, S., J. Nanostructures, 2015, vol. 5, pp. 183–187.
Gurylev, V., Su, C.-Y., and Perng, T.-P., J. Catalysis, 2015, vol. 330, pp. 177–186.
Hua, M., Zhang, S., Pan, B., Zhang, W., Lv, L., and Zhang, Q., J. Hazard. Mat., 2012, vol. 211, pp. 317–331.
Schneider, J., Matsuoka, M., Takeuchi, M., et al., Chem. Rev., 2015, vol. 114, p. 9919–9986.
Baglov, A.V., Denisov, N.M., Borisenko, V.E., et al., Inorg. Mater., 2017, vol. 53, p. 1180.
Sedneva, T.A., Lokshin, E.P., Belyaevskii, A.T., et al., Inorg. Mater., 2008, vol. 44, p. 726.
Wen, J., Li, X., Liu, W., Fang, Y., Xie, J., and Xu, Y., Chinese J. Catalysis, 2015, vol. 36, pp. 2049–2070.
Tian, J., Leng, Y., Zhao, Z., et al., Nano Energy, 2015, vol. 11, pp. 419–427.
Chen, P., Wang, F., Chen, Z.-F., et al., Applied Catalysis B: Environmental, 2017, vol. 204, pp. 250–259.
Sirelkhatim, A., Mahmud, S., Seeni, A., et al., Nano-Micro Let., 2015, vol. 7, pp. 219–242.
Lee, K.M., Lai, C.W., Ngai, K.S., and Juan, J.C., Water Res., 2016, vol. 88, pp. 428–448.
Shanthi, S., Poovaragan, S., Arularasu, M. et al., J. Nanosci. & Nanotech., 2018, vol. 18, pp. 5441–5447.
Shaporev, A.S., Ivanov, V.K., Baranchikov, A.E., et al., Inorg. Mater., 2007, vol. 43, p. 35.
Jana, T., Maji, S., Pal, A., Maiti, R., Dolai, T., and Chatterjee, K., J. Colloid & Inter. Sci., 2016, vol. 480, pp. 9–16.
Ertis, I.F. and Boz, I., International J. Chemical Reactor Engineering, 2017, vol. 15.
Momeni, M., Ghayeb, Y., and Gheibee, S., Ceramics International, 2017, vol. 43, vol. 564–570.
Ofori, F.A., Sheikh, F.A., Appiah-Ntiamoah, R., Yang, X., and Kim, H., Nano-Micro Letters, 2015, vol. 7, pp. 291–297.
Huang, Z.F., Song, J., Pan, L., Zhang, X., Wang, L., and Zou, J.J., Advanced Materials, 2015, vol. 27, pp. 5309–5327.
Sudha, D. and Sivakumar, P., Chemical Engineering and Processing: Process Intensification, 2015, vol. 97, pp. 112–133.
Liu, X., Zeng, J., Yang, H., Zhou, K., and Pan, D., RSC Advances, 2018, vol. 8, pp. 4014–4031.
Ramasami, A.K., Reddy, M., Nithyadharseni, P., Chowdari, B., and Balakrishna, G.R., J. Alloys and Compounds, 2017, vol. 695, pp. 850–858.
Sun, Y., Liang, X., Xiang, H., and Yu, Y., Chinese Chemical Letters, 2017, vol. 28, pp. 2251–2253.
Rajeshwari, S., Kumar, J.S., Rajendrakumar, R., Ponpandian, N., and Thangadurai, P., Mat. Res. Express, 2018, vol. 5, 025507.
Vattikuti, S.P., Reddy, P.A.K., NagaJyothi, P., Shim, J., and Byon, C., J. Alloys and Compounds, 2018.
Aslam, M., Ismail, I.M., Salah, N., Chandrasekaran, S., Qamar, M.T., and Hameed, A., J. Hazard. Mat., 2015, vol. 286, pp. 127–135.
Fei, H.-L., Zhou, H.-J., Wang, J.-G., Sun, P.-C., Ding, D.-T., and Chen, T.-H., Solid State Sci., 2008, vol. 10, pp. 1276–1284.
Li, Y., Kuang, J.-L., Lu, Y., and Cao, W.-B., Acta Metallurgica Sinica (English Letters), 2017, vol. 30, pp. 1017–1026.
Jianhua, L., Rong, Y., and Songmei, L., Rare Metals., 2006, vol. 25, pp. 636–642.
Shanmugam, M., Alsalme, A., Alghamdi, A., and Jayavel, R., ACS Appl. Mat. & Int., 2015, vol. 7, pp. 14905–14911.
Mi, L., Huang, Y., Qin, C., Qin, L., and Seo, H.J., J. Luminescence, 2018, vol. 194, pp. 414–419.
Suresh, R., Giribabu, K., Manigandan, R., et al., J. Alloys and Compounds, 2017, p. 598, 151–160.
Kundu, S., Satpati, B., Kar, T., and Pradhan, S.K., J. Hazard. Mat., 2017, vol. 339, 161–173.
Zengin, Y., and Pozan Soylu G.S., Env. Eng. Sci., vol. 35, pp. 323–332.
Mousavi, M., Kompany, A., Shahtahmasebi, N., and Bagheri-Mohagheghi, M., Advances in Manufacturing, 2013, vol. 1, pp. 320–328.
Mousavi, M. and Yazdi, S.T., Modern Physics Letters B, 2016, vol. 30, 1650151.
Zhu, Y.-N., Zheng, G.-H., Dai, Z.-X., Mu, J.-J., and Yao, Z.-F., J. Mat. Sci. & Techn., 2017, vol. 33, pp. 834–842.
Jia, Z., Kang, J., Zhang, W., et al., Appl. Cat., B: Environmental, 2017, vol. 204, pp. 537–547.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kerli, S., Alver, Ü., Eskalen, H. et al. Structural and Morphological Properties of Boron Doped V2O5 Thin Films: Highly Efficient Photocatalytic Degradation of Methyl Blue. Russ J Appl Chem 92, 304–309 (2019). https://doi.org/10.1134/S1070427219020216
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427219020216