Abstract
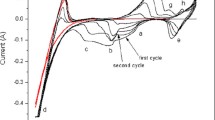
Crude vanadium-containing dump slimes can be used as a raw material for preparing lithium vanadium oxides suitable as cathode materials for lithium-ion chemical current sources. A simple procedure for preparing LiV3O8 from the slime was suggested. The synthesized compound was characterized by X-ray diffraction, Raman spectroscopy, scanning electron microscopy, and synchronous thermal analysis. The material prepared from vanadium-containing dump slimes has uniform microstructure and high electronic conductivity of the order of 1.4 × 10–2 S cm–1; it is thermally stable in the interval 30–550°С and capable of intercalation/deintercalation of lithium ions in its structure in the course of cycling in lithium-ion current sources.
Similar content being viewed by others
References
Enjalbert, R. and Galy, J., Acta Crystallogr., Sect. C, 1986, vol. 42, pp. 1467–1469.
Balog, P., Orosel, D., Cancarevic, Z., Schon, C., and Jansen, M.J., J. Alloys Compd., 2007, vol. 429, pp. 87–98.
Whittingham, M.S., Chem. Rev., 2004, vol. 104, pp. 4271–4302.
Murphy, D.W., Christian, P.A., Disalvo, F.J., and Waszczak, J.V., Inorg. Chem., 1979, vol. 18, pp. 2800–2803.
Whittingham, M.S., J. Electrochem. Soc., 1976, vol. 126, pp. 315–320.
Wiesener, K., Schneider, W., Ilic, D., Steger, E., Hallmeier, K.H., and Brackmann, E., J. Power Sources, 1987, vol. 20, pp. 157–164.
Delmas, C., Cognac-Auradou, H., Cocciantelli, J.M., Ménétrier, M., and Doumerc, J.P., Solid State Ionics, 1994, vol. 69, pp. 257–264.
Bates, J.B., Gruzalski, G.R., Dudney, N.J., Luck, C.F., and Yu, X., Solid State Ionics, 1994, vols. 70–71, pp. 619–628.
Cocciantelli, J.M., Doumerc, J.P., Pouchard, M., Broussely, M., and Labat, J., J. Power Sources, 1991, vol. 34, pp. 103–111.
Gong, H. and Qian, Y., Chin. J. Chem. Phys., 2013, vol. 26, p. 597.
Whittingham, M.S., Chem. Rev., 2004, vol. 104, pp. 4271–4301.
Ivanov-Shits, A.K. and Dem’yanets, L.N., Priroda, 2003 no. 12, pp. 35–43.
Sanchez, C., Livage, J., and Lucazeau, G., J. Raman Spectrosc., 1982, vol. 12, pp. 68–72.
Zhou, B. and He, D., J. Raman Spectrosc., 2008, vol. 39, pp. 1475–1481.
Baddour-Hadjean, R., Smirnov, M.B., Smirnov, K.S., Kazimirov, V.Yu., Gallardo-Amores. J.M., Amador, U., Arroyo-de Dompablo, M.E., and Pereira-Ramos, J.P., Inorg. Chem., 2012, vol. 51, pp. 3194–3201.
Wadsley, A.D., Acta Crystallogr., 1957, vol. 10, p. 261.
Zhang, X. and Frech, R., Electrochim. Acta, 1998, vol. 43, pp. 861–868.
Brooker, M.H. and Bates, J.B., J. Chem. Phys., 1971, vol. 54, pp. 4788–4796.
Slobodin, B.V., Vanadaty s-elementov (s Element Vanadates), Yekaterinburg: Ural’skoe Otdel. Ross. Akad. Nauk, 2008, p. 133.
Wang, C. and Hong, J., Electrochem. Solid-State Lett., 2007, vol. 10, pp. A65–A69.
Guan, J. and Liu, M., Solid State Ionics, 1998, vol. 110, pp. 21–28.
Wolfenstine, J. and Allen, J., J. Power Sources, 2008, vol. 180, pp. 582–585.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.S. Shchelkanova, G.Sh. Shekhtman, A.A. Pankratov, 2018, published in Zhurnal Prikladnoi Khimii, 2018, Vol. 91, No. 11, pp. 1595−1601.
Rights and permissions
About this article
Cite this article
Shchelkanova, M.S., Shekhtman, G.S. & Pankratov, A.A. Use of Vanadium-Containing Slime for Preparing Cathodes for Lithium-Ion Current Sources. Russ J Appl Chem 91, 1799–1804 (2018). https://doi.org/10.1134/S1070427218110095
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427218110095