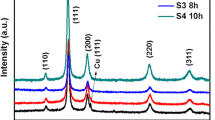
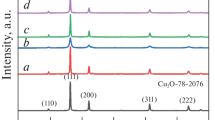
Abstract
new material of Cu(OH)2 nanostructures was prepared using cupric nitrate and sodium hydroxide as raw materials by the chemical precipitation method. The Cu(OH)2 nanostructures were characterized by scanning electron microscope, transmission electron microscopy, infrared spectrometer, and X-ray diffractometer. The results showed that the Cu(OH)2 nanostructures exhibited excellent uniform and dispersion at 40°C. A series of factors was investigated to effect the photocatalytic efficiency of methyl orange (MO), such as the concentration of Cu(OH)2 nanostructures, the reaction time of the Cu(OH)2 nanostructures, the initial concentration of MO, and so on. As a result, the Cu(OH)2 nanostructures exhibited excellent photocatalytic efficiency with the concentration of 20 mg L–1 Cu(OH)2 nanostructures, the initial concentration of MO was 15 mg L–1 and the stirring time was 70 min.
Similar content being viewed by others
References
Mittal, A., Mittal, J., Malviya, A., Kaur, D., and Gupta, V.K., J. Colloid Interface Sci., 2010, vol. 342, p. 518.
Mittal, A., Mittal, J., Malviya, A., and Gupta, V.K., J. Colloid Interface Sci., 2009, vol. 340, p. 16.
Forgacs, E., Cserhati, T., and Oros, G., Environ. Int., 2004, vol. 30, p. 953.
Saleh, T.A. and Gupta, V.K., Adv. Colloid Interface Sci., 2014, vol. 211, p. 93.
Scott, J.P. and Ollis, D.F., Environ. Prog., 1995, vol. 14, p. 88.
Dey, A.N. and Sullivan, B.P., J. Electrochem. Soc., 1970, vol. 117, p. 222.
Petushkova, J.P. and Lyalikova, N.N., Stud. Conserv., 1986, vol. 31, p. 65.
Sholkovitz, E.R., Geochim. Cosmochim. Acta, 1976, vol. 40, p. 831.
Turchi, C.S. and Ollis, D.F., J. Catal., 1990, vol. 122, p. 178.
Gupta, V.K., Ali, I., Saleh, T.A., and Nayak, A., S. RSC Adv., 2012, vol. 2, p. 6380.
Hoffmann, M.R., Chem. Rev., 1995, vol. 95, p. 69.
Fujishima, A., Zhang, X., and Tryk, D., Int. J. Hydrogen Energy, 2007, vol. 32 p. 2664.
Ochiai, T. and Fujishima, A., J. Photochem. Photobiol. C Photochem. Rev., 2012, vol. 13, p. 247.
Zhang, N., J. Catal., 2013, vol. 303, p. 60.
Shen, X., Atmos. Environ., 2013, vol. 68, p. 297.
Liu, X., Cheng, Y., Li, X., and Dong, J., Appl. Surf Sci., 2017, doi: https://doi.Org/10.1016/ j.apsusc.2017.12.201, http:// muchong.com/bbs/journal.php?view=detail&jid=773.
Sun, P., You, L., Wang, D., Sun, Y., Ma, J., and Lu, G., Sens. Actuators B Chem., 2011, vol. 156, p. 368.
Zhang, Y. and Lu, J., Cryst. Growth Des., 2008, vol. 8, p. 2101.
Shinde, S.K., Dubal, D.P., Ghodake, G.S., Kim, D.Y., and Fulari, V.J., J. Electroanal. Chem., 2014, vol. 732, p. 80.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Li, K., Lu, J., Zheng, X. et al. Cu(OH)2 Nanostructures for Dynamic Photodegradation of Methyl Orange under Visible Light. Russ J Appl Chem 91, 1345–1352 (2018). https://doi.org/10.1134/S107042721808013X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042721808013X