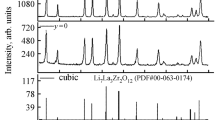
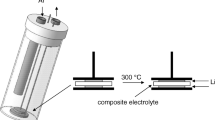
Abstract
Cyclability of the Li|Li7La3Zr2O12 interface was tested by voltammetry under externally applied potential difference. It was found that the solid electrolyte synthesized in the study contains a minor amount of an impurity in the form of lithium carbonate. This impurity forms, when brought in contact with metallic lithium, carbon that pierces the whole volume of the ceramic separator and produces a channel for a flow of electrons through the material, which leads to a poor cyclability of the solid electrolyte. A possible way to solve the given problem is via a purposeful replacement of the carbonate in the intergrain space of Li7La3Zr2O12 with another crystalline or glassy plasticizer that possesses an acceptable unipolar lithium conductivity (no less than 10–6 S cm–1) and forms, when brought in contact with metallic lithium, no electrically conducting compound or a compound capable of reversibly intercalating/deintercalating lithium.
Similar content being viewed by others
References
Etacheri, V., Marom, R., Elazari, R., et al., Energy Environ. Sci., 2011, vol. 4, pp. 3243–3262.
Burmakin, E.I., Tverdye elektrolity s provodimost’yu po kationam shchelochnykh metallov (Solid Electrolytes with Conductivity by Alkali Metal Cations), Moscow: Nauka, 1992.
Ramakumar, S., Deviannapoorani, C., Dhivya, L., et al., Prog. Mater. Sci., 2017, vol. 88, pp. 325–411.
Zeier, W.G., Royal Soc. Chem., 2014, vol. 43, pp. 16133–16138.
Buschmann, H., Dölle, J., Berendts, S., et al., Phys. Chem. Chem. Phys., 2011, vol. 13, no. 43, pp. 19378–19392.
Murugan, R., Thangadurai, V., and Weppner, W., Angew. Chem. Int. Ed., 2007, vol. 46, pp. 7778–7781.
Il’ina, E.A., Andreev, O.L., Antonov, B.D., et al., J. Power Sources, 2012, vol. 201, pp. 169–173.
Awaka, J., Kijima, N., and Hayakawa, H., J. Solid State Chem., 2009, vol. 182, pp. 2046–2052.
Geiger, C.A., Alekseev, E., Lazic, B., et al., Inorg. Chem., 2011, vol. 50, pp. 1089–1097.
Cheng, L., Crumlin, E.J., Chen, W., et al., Phys. Chem. Chem. Phys., 2014, vol. 16, pp. 18294–18300.
Il’ina, E.A., Andreev, O.L., Antonov, B.D., et al., Mater. Res. Bull., 2014, vol. 53, pp. 32–37.
Han, X., Gong, Y., Fu, K., et al., Nat. Mater., 2016, vol. 16, pp. 572–580.
Damaskin, B.B., Printsipy sovremennykh metodov izucheniya elektrokhimicheskikh reaktsii (Principles of Modern Methods for Study of Electrochemical Reactions), Moscow: Mosk. Gos. Univ., 1965.
Damaskin, B.B., Petrii, O.A., and Tsirlina, G.A., Elektrokhimiya (Electrochemistry), Moscow: Khimiya, KolosS, 2006, 2nd ed.
Electroanalytical Methods. Guide to Experiments and Applications, Scholz, F., Ed., Springer–Verlag, Berlin, 2010.
O’Callaghan, M.P. and Cussen, E.J., Solid State Sci., 2008, vol. 39, pp. 390–395.
Cussen, E.J., Chem. Commun., 2006, vol. 4, pp. 412–413.
Handbook of Battery Materials, Besenhard, R., Ed., Wiley–VCH Verlag, 1999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.V. Druzhinin, P.Yu. Shevelin, E.A. Il’ina, 2018, published in Zhurnal Prikladnoi Khimii, 2018, Vol. 91, No. 1, pp. 70−76.
Rights and permissions
About this article
Cite this article
Druzhinin, K.V., Shevelin, P.Y. & Il’ina, E.A. Cycling Performance at Li7La3Zr2O12|Li Interface. Russ J Appl Chem 91, 63–69 (2018). https://doi.org/10.1134/S107042721801010X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042721801010X