Abstract
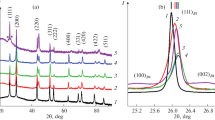
Working conditions and the concentration regions of the joint precipitation of PbS and CdS at which substitution solid solutions Cd x Pb1–x can be formed were determined by calculation of ionic equilibria in the citrate-ammonia reaction mixture at 298 and 353 K with consideration for the conversion fractions of lead and cadmium sulfides into the corresponding sulfides. The hydrochemical precipitation onto glass-ceramic substrates was used to obtain Cd x Pb1–x S (0 < x ≤ 0.149) solid solution films with thicknesses of 0.5 to 1.7 μm and high supersaturation with the substituting component. All the films crystallize to form the B1 structure. The phase and elemental compositions and morphological specific features of the films were studied. It was shown that the thickness of the deposited layers is most strongly affected by the process temperature, ammonium hydroxide concentration, and relative amounts of the metal salts in the reaction mixture. It was found that there are oxygen and chlorine in the Cd x Pb1–x S solid solutions, and the distribution of these elements across the layer thickness was determined, with the layer-by-layer distribution of chlorine having a pronounced oscillatory nature. It was shown that, as the chemical precipitation temperature is raised, the content of CdS in the substitution solid solutions grows exponentially. The activation energies Ea.ic of the lead and cadmium interchange in the PbS crystal lattice were found to be, depending on the initial concentration of the lead salt, 75.3, 42.8, and 22.2 kJ mol–1.
Similar content being viewed by others
References
Patricia L. Nichols, Zhicheng Liu, Leijun Yin, et al., Nano Lett., 2015, no. 15, pp. 909–916.
Hernadez–Borja J., Vorobiev Y. V., and Ramirez–Bon, J., Solar Energy Mater. Solar Cells, 2011, vol. 95, pp. 1882–1888.
Markov, V.F., Maskaeva, L.N., and Polikarpova, Yu.S., Butlerov Soobshch., 2006, vol. 8, no. 1, pp. 54–61.
Maskaeva, L.N., Markov, V.F., Porkhachev, M.Yu., and Mokrousova, O.A., Pozharovzryvobezopasnost’, 2015, vol. 24, no. 9, pp. 67–73.
Chattraki, A.N., Kamble, S.S., and Deshmukh, L.P., Mater. Lett., 2012, no. 67, pp. 39–41.
Obaid, A.S., Mahdi, M.A., Hassan, Z., and Bououdina, M., Superlattices Microstruct., 2012, no. 52, p. 816.
Guo-Long Tan, Limin Liu, and Weibing Wu, AIP Adv., 2014, no. 4, p. 067107.
Urusov, V.S., Tauson, V.L., and Akimov, V.V., Geokhimiya tverdogo tela (Solid State Geochemistry), Moscow: GEOS, 1997.
Eman M. Nasir and Iqbal S. Naji, Australian J. Basic Appl. Sci., 2015, vol. 9, no. 20, pp. 364–371.
Rokakh, A.G., Bilenko, D.I., Shishkin, M.I., et al., Semiconductors, 2014, vol. 48, no. 12, pp. 1562–1566.
Anbarasi, M., Nagarethinam, V.S., Baskaran, R., and Narasimman, V., Pacific Sci. Rev. A: Natural Sci. Eng., 2016, vol. 18, no. 1, pp. 72–77.
Rabinovich, E., Wachtel, E., and Hodes, G., Thin Solid Films, 2008. 517, pp. 737–744.
Shelimova, L.E., Tomashik, V.N., and Grytsiv, V.I., Diagrammy sostoyaniya v poluprovodnikovom materialovedenii (sistemy na osnove khal’kogenidov Si, Ge, Sn, Pb) [Constitution Diagrams in Semiconductor Materials Science (Systems Based on Si, Ge, Sn, Pb Chalcogenides), Moscow: Nauka, 1991.
Markov, V.F., Maskaeva, L.N., and Ivanov, P.N., Gidrokhimicheskoe osazhdenie plenok sul’fidov metallov: modelirovanie i eksperiment (Hydrochemical Deposition of Metal Sulfide Films: Simulation and Experiment), Yekaterinburg: Ural. Otd. Ross. Akad. Nauk, 2006.
Obaid, A.S., Hassan, Z., Mahdi, M.A., and Bououdina, M., Sol. Energy, 2013, vol. 89, pp. 143–151.
Barote, M.A., Kamble, S.S., Deshmukh, L.P., and Masumdar, E.U., Ceram. Int., 2013, vol. 39, pp. 1463–1467.
Suryavanshi, K.E., Dhake, R.B., Patil, A.M., et al., Int. J. Adv. Res., 2014, vol. 2, no. 6, pp. 491–493.
Suryavanshi, K.E., Patil, A.M., and Dhake, R.B., Der Chem. Sinica, 2015, vol. 6, no. 6, pp. 59–65.
Deo, S.R., Singh, A.K., Deshmukh, L., et al., Optik–Int. J. Light Electron Optics, 2015, vol. 126, no. 20, pp. 2311–2317.
Suryavanshi, K.E., Dhake, R.B., and Patil, A.M., Int. J. Adv. Sci. Tech. Res., 2014, vol. 2, no. 4, pp. 858–861.
Ahmad, S.M., Kasim, S.J., and Latif, L.A., Jordan J. Phys., 2016, vol. 9, no. 2, pp. 113–122.
Rodriguez-Carvajal, J., Physica B, 1993, vol. 192, pp. 55–64.
Maskaeva, L.N., Glukhova, I.A., Markov, V.F., et al., Russ. J. Appl. Chem., 2016, vol. 89, no. 12, pp. 1939–1947.
Martell, A.E. and Hancock, R.D., Metal Complexes in Aqueous Solutions, New York: Plenum Press, 1996.
Volkov, B.A., Ryabova, L.I., and Khokhlov, D.R., Uspekhi Fiz. Nauk, 2002, vol. 172, no. 8, pp. 875–906.
Maskaeva, L.N., Markov, V.F., and Gusev, A.I., Russ. J. Inorg. Chem., 2004, vol. 49, no. 7, pp. 971–977.
Markov, V.F., Maskaeva, L.N., and Kitaev, G.A., Russ. J. Appl. Chem., 2000, vol. 73, no. 8, pp. 1328–1331.
Maskaeva, L.N., Markov, V.F., Forostyanaya, N.A., et al., Zh. Obshch. Khim., 2016, vol. 86, no. 10, pp. 1624–1632.
Maskaeva, L.N., Markov, V.F., Vaganova, I.V., and Forostyanaya, N.A., Butlerov Soobshch., 2017, vol. 49, no. 3, pp. 50–59.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.N. Maskaeva, I.V. Vaganova, V.F. Markov, V.I. Voronin, 2017, published in Zhurnal Prikladnoi Khimii, 2017, Vol. 90, No. 5, pp. 553−563.
Rights and permissions
About this article
Cite this article
Maskaeva, L.N., Vaganova, I.V., Markov, V.F. et al. Films of supersaturated Cd x Pb1–x S solid solutions: Composition prognostication, chemical synthesis, microstructure. Russ J Appl Chem 90, 691–700 (2017). https://doi.org/10.1134/S1070427217050044
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427217050044