Abstract
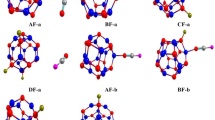
The adsorption of the 2,3,7,8-tetrachlorodibenzodioxin (TCDD) molecule on the B12N12 nanocage (B12N12-NC) was studied by M06-2X/6-31++G** method. There are three sites for TCDD adsorption on B12N12-NC. The B–B atom pair in six-membered rings (B(6MR)–B(6MR)) of B12N12-NC is the preferable adsorption site. When TCDD approaches the B12N12 nanocage, electronic exchange between them occurs, and TCDD is converted to 3,4-dichlorophenol, 3-chloroprop-2-en-1-ol, and 1-chloroprop-1-ene. The HOMO/LUMO energy, energy gaps (E g), thermodynamic properties, and structural deformation are calculated by DFT methods. The lowest value of E g (3.796 eV) was obtained for TS-3 (the first transition state of conversion of intermediate 3,4-dichlorophenol to 3-chloroprop-2-en-1-ol and 1-chloroprop-1-ene). The Gibbs free energy and heat of reactions are negative; therefore, these reactions are favorable and spontaneous and make B12N12-NC suitable as nanosensor for TCDD detection and reduction.
Similar content being viewed by others
References
Wang, B.J. et al., J. Hazard. Mater., 2010, vol. 161, pp. 800–807.
Mousavi, M., Taymouri, A., and Ghaffarian, V., Int. J. Environ. Sci. Technol., 2009, vol. 6, no. 3, pp. 499–508.
Esrafili, M.D. and Nurazar, R., Surf. Sci., 2014, vol. 626, pp. 44–48.
Momeniha, F., Nabizadeh, R., Hassanvand, M.S., et al., Iran. Bull. Environ. Contam. Toxicol., 2011, vol. 87, no. 6, pp. 708–712.
Gharib, A., Vojdanifard, L., Noroozi Pesyan, N., et al., Bulg. Chem. Commun., 2014, vol. 46, no. 4, pp. 667–679.
Jensen, F. and Toftlund, H., Chem. Phys. Lett., 1993, vol. 201, nos. 1–4, pp. 89–96.
Hosseini, M., Ghozatloo, A., and Shariaty-Niassar, M., J. Nanostruct. Chem., 2015, vol. 5, no. 2, pp. 219–226.
Noei, M., Ebrahimikia, M., Saghapour, Y., et al., J. Nanostruct. Chem., 2015, vol. 5, no. 2, pp. 213–217.
Mazhdi, M., Saydi, J., and Mazhdi, F., Int. J. Bio-Inorg. Hybr. Nanomater., 2013, vol. 2, no. 1, pp. 295–302.
Rajeshkumar, S., Ponnanikajamideen, M., Malarkodi, C., et al., J. Nanostruct. Chem., 2014, vol. 4, paper 96, pp. 1–9.
Yourdkhani, S., Korona, T., and Hadipour, N.L., J. Phys. Chem. A, 2015, vol. 119, no. 24, pp. 6446–6467.
Douglas, L., J. Phys. Chem. A, 2000, vol. 104, no. 15, pp. 3364–3366.
Noei, M., Asadi, H., Salari, A.A., and Hosseini Mahjoob, S.M.R., Indian J. Fundam. Appl. Life Sci., 2014, vol. 4, no. 2, pp. 679–-685.
Beheshtian, J., Peyghan, A.A., and Bagheri, Z., Sens. Actuators B: Chemical, 2012, vols. 171–172, pp. 846–852.
Beheshtiana, J., Kamfiroozi, M., Bagheri, Z., and Ahmadi Peyghan, A., Chin. J. Chem. Phys., 2012, vol. 25, no. 1, pp. 60–64.
Baei, M.T., Ahmadi Peyghan, A., and Bagheri, Z., Bull. Korean Chem. Soc., 2012, vol. 33, no. 10, pp. 3338–3342.
Ahmadi Peyghan, A. and Soleymanabadi, H., Curr. Sci., 2015, vol. 108, no. 10, pp. 1910–1914.
Baei, M.T., Comput. Theor. Chem., 2013, vol. 1024, pp. 28–33.
Seifert, G., Fowler, R.W., Mitchell, D., et al., Chem. Phys. Lett., 1997, vol. 268, pp. 352–358.
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., et al., J. Comput. Chem., 1993, vol. 14, pp. 1347–1363.
Oku, T., Nishiwaki, A., and Narita, I., Sci. Technol. Adv. Mater., 2004, vol. 5, pp. 635–638.
Kaya, E.N., Tuncel, S., Basova, T.V., et al., Sens._Actuators B, 2014, vol. 199, pp. 277–283.
Bahrami, A., Seidi, S., Baheri, T., and Aghamohammadi, M., Superlatt. Microstruct., 2013, vol. 64, pp. 265–273.
Esrafili, M.D. and Nurazar, R., Superlatt. Microstruct., 2014, vol. 67, pp. 54–60.
Walker, M., Harvey, A.J.A., Sen, A., and Dessent, C.E.H., J. Phys. Chem. A, 2013, vol. 117, no. 47, pp. 12590–12600.
Lynam, M.M., Kuty, M., Damborsky, J., et al., Environ. Toxicol. Chem., 1998, vol. 17, no. 6, pp. 988–997.
Ahmadi, R. and Pirahan-Foroush, M., Ann. Med. Health Sci. Res., 2014, vol. 12, pp. 39-43.
Shakerzadeh, E., Barazesh, N., and Zargar Talebi, S., Superlatt. Microstruct., 2014, vol. 76, pp. 264–276.
Baei, M.T., Soltani, A., Torabi, P., and Hashemian, S., Monatsh. Chem., 2015, vol. 146, no. 6, pp. 891–896.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the author in English.
Rights and permissions
About this article
Cite this article
Mahdavian, L. A study of B12N12 nanocage as potential sensor for detection and reduction of 2,3,7,8-tetrachlorodibenzodioxin. Russ J Appl Chem 89, 1528–1535 (2016). https://doi.org/10.1134/S1070427216090226
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427216090226