Abstract
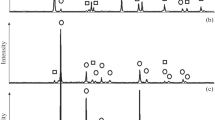
Titania powders with anatase structure and crystallite size ranged from 20 to 27 nm were produced from titanium hydroxide by hydrothermal synthesis. The nanopowders and the initial hydroxide were isothermally heat-treated in a hydrogen flow at temperatures of 00–1000°C for times ranging from 1 to 27.5 h. It was found that the thermal treatment of titanium hydroxide in a reducing atmosphere at 800–900°C yields TiO2 with rutile structure. An unusual thermal stability of the metastable structure of anatase was observed in the case of nanocrystalline TiO2 powders up to a temperature of 900°C at which the anatase–rutile phase transition is observed. It was found that the nonstoichiometry of rutile and anatase powders changes upon a thermal treatment in a hydrogen flow. The heat-treatment in a hydrogen flow at 1000°C for 1 h was shown to lead to the formation of nanoparticles of the Magneli phase with the composition corresponding to Ti9O17 irrespective of the chemical nature of the precursor. The increase of the heat-treatment time at this temperature leads to the appearing of the Magneli phases with the compositions corresponding to Ti8O15 and Ti9O17 and Ti2O3 and Ti3O5 suboxides in addition to the Ti9O17 main phase.
Similar content being viewed by others
References
Chen, D., Chen, C., Baiyee, Z.M., et al., Chem. Rev., 2015, vol. 115, no. 18, pp. 9869–9921.
Kwon, D.-H., Kim, K.M., Jang, J.H., et al., Nat. Nanotechnol., 2010, vol. 5, no. 2, pp. 148–153.
Gusev, A.I., Rempel, A.A., and Magerl, A.J., Disorder and Order in Strongly Nonstoichiometric Compounds: Transition Metal Carbides, Nitrides and Oxides, Berlin: Springer-Verlag, 2001, p. 617.
Cava, R.J., Batlogg, B., Krajewski, J.J., et al., Phys. Rev. B, 1991, vol. 44, no. 13, pp. 6973–6981.
Smith, J.R., Clarke, R.L., and Walsh, F.C., J. Appl. Electrochem., 1998, vol. 28, no. 10, pp. 1021–1033.
Han, W.-Q. and Wang, X.-L., Appl. Phys. Lett., 2010, vol. 97, pp. 243104–243107.
Styles, M.J., In Situ Studies of the Structure and Oxidation of Magnŭli Phase Electrodes and Their Application in Molten Salt Electrolysis: PhD thesis, Eng.-Mech. Eng., The University of Melbourne, 2012, p. 343.
Teleki, A. and Pratsisinis, S.E., Phys. Chem. Chem. Phys., 2009, vol. 11, pp. 3724–3747.
Wang, Y., Qin, Y., Li, G., et al., J. Cryst. Growth, 2005, vol. 282, pp. 402–406.
Hayfield, P.C.S., Development of a New Material: Ti4O7 Monolithic Ebonex® Ceramic, Cambridge: Royal Society of Chemistry, 2002, p. 97.
Qian, S. and Mao, J., J. Mater. Sci.: Mater Electron., 2015, vol. 26, pp. 5166–5172.
Bullard, D.E. and Lynch, D.C., Metall. Mater. Trans. B, 1997, vol. 28, pp. 1069–1080.
Palmer, R.A., Doan, T.M., Lloyd, P.G., et al., Plasma Chem. Plasma Process., 2002, vol. 22, no. 3, pp. 335–350.
Zhang, Y.-W., Ding, W.-Z., and Xu, K.-D., Trans. Nonferrous Met. Soc. China, 2005, vol. 15, no. 3, pp. 594–599.
Binnewies, M., Glaum, R., Schmidt, M., and Schmidt, P., Z. Anorg. Allg. Chem., 2013, vol. 639, no. 2, pp. 219–229.
Hauf, C., Kniep, R., and Pfaff, G., J. Mater. Sci., 1999, vol. 34, no. 6, pp. 1287–1292.
Khader, M.M., Kheiri, M.-N., El-Anadouli, B.E., and Ateya, B.G., Phys. Chem., 1993, vol. 97, pp. 6074–6077.
Lynch, D.C. and Bullard, D.E., Metall. Mater. Trans. B, 1997, vol. 28, pp. 447–453.
Okamoto, H., JPED, 2011, vol. 32, no. 5, pp. 473–474.
Kolen’ko, Yu.V., Burukhin, A.A., Churagulov, B.R., et al., J. Inorg. Chem., 2002, vol. 47, no. 11, pp. 1609–1615.
Lutterotti, L., Nucl. Instrum. Meth. (B), 2010, vol. 268, pp. 334–340.
Ida, T., Shimazaaki, S., Hibino, H., and Toraya, H., J. Appl. Cryst., 2003, vol. 36, pp. 1107–1115.
Depero, L.E., Sangaletti, L., Allieri, B., et al., J. Cryst. Growth, 1999, vols. 198–199, pp. 516–520.
Hanaor, D.A.H. and Sorrell, C.C., J. Mater. Sci., 2011, vol. 46, pp. 855–874.
Satoh, N., Nakashima, T., and Yamamoto, K., Sci. Rep., 2013, vol. 3, Article no. 1959. http://dx.doi.org/10.1038/srep01959.
Nolan, N., Seery, M., and Pillai, S., J. Phys. Chem. C, 2009, vol. 113, pp. 16151–16157.
Hashimoto, K., Irie, H., and Fujishima, A., Jpn. J. Appl. Phys., 2005, vol. 44, no. 12, pp. 8269–8285.
Katzke, H. and Schlğl, R., J. Acta Cryst. B, 2003, vol. 59, pp. 456–462.
Syarif, D.G., Miyashita, A., Yamaki, T., et al., Appl. Surf. Sci., 2002, vol. 193, pp. 287–292.
Byrappa, K. and Adschiri, T., Prog. Cryst. Growth Charact. Mater., 2007, vol. 53, no. 2, pp. 117–166.
Makinson, J.D., Lee, J.S., Magner, S.H., et al., Adv. X-Ray Anal., 2000, vol. 42, pp. 407–411.
Jenkins, R. and Snyder, R.L., Hoboken, New York: John Wiley & Sons, Inc., 1996, p. 432.
Melikhov, I.V., Ros. Khim. Zh. (Zh. Ros. Khim. O–va im. D. I. Mendeleeva), 2002, vol. 46, no. 5, pp. 7–14.
Ranade, M.R., Navrotsky, A., Zhang, H.Z., et al., Proc. Natl. Acad. Sci. U.S.A., 2002, vol. 99, pp. 6476–6481.
Uvarov, N.F. and Boldyrev, V.V., Rus. Chem. Rev., 2001, vol. 70, no. 4, pp. 265–284.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.K. Vasilevskaia, V.I. Popkov, A.A. Valeeva, A.A. Rempel’, 2016, published in Zhurnal Prikladnoi Khimii, 2016, Vol. 89, No. 8, pp. 961−970.
Rights and permissions
About this article
Cite this article
Vasilevskaia, A.K., Popkov, V.I., Valeeva, A.A. et al. Formation of nonstoichiometric titanium oxides nanoparticles Ti n O2n–1 upon heat-treatments of titanium hydroxide and anatase nanoparticles in a hydrogen flow. Russ J Appl Chem 89, 1211–1220 (2016). https://doi.org/10.1134/S1070427216080012
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427216080012