Abstract
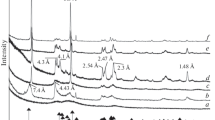
Method for synthesis of calcium aluminate and sorbents based on this compound was developed on its basis of the sol-gel technology. The method makes it possible to obtain the target product with specific surface areas in the range from 7 to 120 m2 g–1. The compounds obtained were characterized by scanning electron microscopy, X-ray phase analysis, and X-ray fluorescence microanalysis. It was found that the main phase of the sorbents is rhombohedral calcium monoaluminate CaAl2O4. The method of Hammett indicator adsorption was used to determine the content of active centers on the surface of the sorbents and the distribution of these centers over ionization constants. It was shown that the surface characteristics of the sorbents depend on their synthesis method. The retention parameters, polarities, and thermodynamic characteristics were determined for organic compounds of various classes. The possibility of using calcium aluminate modified with sodium chloride for gas-chromatographic separation of aliphatic and aromatic hydrocarbons and carbonyl compounds was demonstrated.
Similar content being viewed by others
References
Van den Eeckhout, K., Smet, P.F., and Poelman, D., Materials, 2010, vol. 3, no. 4, pp. 2536–2566.
Choi, S.W. and Hong, S.H., Mater. Sci. Eng. B, 2010, vol. 171, nos. 1–3, pp. 69–72.
Hafiane, Y.El, Smith, A., Abouliatim, Y., et al., J. Eur. Ceram. Soc., 2014, vol. 34, pp. 1017–1023.
Li Jin-hong, Cai Bi-ya, Feng Wu-wei, et al., Ceram. Int., 2013, vol. 39, pp. 8393–8400.
Shiri, S., Abbasi, M.H., Monshi, A., and Karimzadeh, F., Adv. Powder Technol., 2014, vol. 25, pp. 338–341.
Avci, N., Korthout, K., Newton, M.A., et al., Opt. Mater. Express, 2012, vol. 2, no. 3, pp. 321–330.
Mukhopadhyay, S. and Dutta, S., Ceram. Int., 2012, vol. 38, pp. 4997–5006.
Zhang Yin, Chen Jie, Chuanyan Xu, et al., Physica B, 2015, vol. 472, pp. 6–10.
Janakovaa, S., Salavcova, L., Renaudin, G., et al., J. Phys. Chem. Solids, 2007, vol. 68. pp. 1147–1151.
Selyunina, L.A., Mishenina, L.N., Slizhov, Yu.G., and Kozik, V.V., Russ. J. Inorg. Chem., 2013, vol. 58, no. 4, pp. 450–455.
Zakharova, N.V., Sychev, M.M., Korsakov, V.G., and Myakin, S.V., Kondens. Sredy Mezhfaznye Granitsy, 2011, vol. 13, no. 1, pp. 56–62.
Khimiya privitykh poverkhnostnykh soedinenii (Chemistry of Grafted Surface Compounds), Lisichkin, G.V., Ed., Moscow: Fizmatlit, 2003.
Lukás, J., Svec, F., Votavová, E., and Kálal, J., J. Chromatogr., 1978, vol. 153, pp. 373–380.
Rabinovich, V.A. and Khavin, Z.Ya., Kratkii khimicheskii spravochnik (Concise Chemical Reference Book), Leningrad: Khimiya, 1991.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.A. Selyunina, L.N. Mishenina, K.P. Mashkova, Yu.G. Slizhov 2016, published in Zhurnal Prikladnoi Khimii, 2016, Vol. 89, No. 3, pp. 341−347.
Rights and permissions
About this article
Cite this article
Selyunina, L.A., Mishenina, L.N., Mashkova, K.P. et al. Sol-gel synthesis and sorption properties of calcium monoaluminate. Russ J Appl Chem 89, 394–399 (2016). https://doi.org/10.1134/S1070427216030083
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427216030083