Abstract
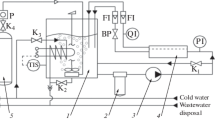
Kinetics of reverse osmosis desalination of aqueous sodium sulfanilate is studied on a laboratory pressure filter unit, in relation to the solution concentration, temperature, and pressure in the intermembrane channel.
Similar content being viewed by others
References
Lazarev, S.I., Korobov, V.B., Kliot, M.B., and Pirogov, P.A., Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 1993, no. 6, pp. 79–80.
Dytnerskii, Yu.I., Baromembrannye protsessy: Teoriya i raschet (Baromembrane Processes: Theory and Calculations), Moscow: Khimiya, 1986.
Hwang, S.-T. and Kammermeyer, K., Membanes in Separations, New York: Wiley, 1975.
Karelin, F.N., Obessolivanie vody obratnym osmosom (Water Desalination by Reverse Osmosis), Moscow: Stroiizdat, 1988.
Bryk, M.T. and Tsapyuk, E.A., Ul’trafil’tratsiya (Ultrafiltration), Kiev: Naukova Dumka, 1989.
Deryagin, B.V., Churaev, N.V., Martynov, G.A., et al., Khim. Tekhnol. Vody, 1981, vol. 3, no. 2, pp. 99–104.
Sourirajan, S., Pure Appl. Chem., 1978, vol. 50, pp. 593–615.
Koganovskii, A.M., Levchenko, T.M., Kirichenko, V.A., et al., Adsorbtsiya rastvorennykh veshchestv (Adsorption of Solutes), Kiev: Naukova Dumka, 1977.
Tone, S., Shinokhara, K., Igorashi, Y., and Otake, T., J. Membrane Sci., 1984, vol. 19, pp. 195–208.
Deryagin, B.V., Churaev, N.V., and Muller, V.M., Poverkhnostnye sily (Surface Forces), Moscow: Nauka, 1985.
Reid, R.F., Properties and Reactions of Bonds in Organic Molecules, London: Longmans, 1968.
Churaev, N.V., Fizikokhimiya protsessov massoperenosa v kapillyarno-poristykh telakh (Physical Chemistry of Mass Transfer in Capillary-Porous Bodies), Moscow: Khimiya, 1990.
Author information
Authors and Affiliations
Additional information
Original Russian Text © S.I. Lazarev, A.S. Gorbachev, V.A. Shaposhnik, V.M. Stamov, 2006, published in Zhurnal Prikladnoi Khimii, 2006, Vol. 79, No. 5, pp. 747–751.
Rights and permissions
About this article
Cite this article
Lazarev, S.I., Gorbachev, A.S., Shaposhnik, V.A. et al. Kinetics of reverse osmosis desalination of aqueous sodium sulfanilate. Russ J Appl Chem 79, 736–740 (2006). https://doi.org/10.1134/S1070427206050089
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1134/S1070427206050089