Abstract
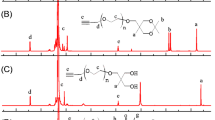
irst-generation dendrimers containing thiacalix[4]arene in cone and 1,3-alternate conformations as a core and hydroxyl groups as terminal moieties have been synthesized. It has been shown that self-assembly of the dendrimers with the macrocyclic core in cone conformation and terminal amidoethanol groups in an aqueous solution leads to the formation of stable monodisperse supramolecular system consisting of the nanoparticles with hydrodynamic diameter of 11.4±0.1 nm.


Similar content being viewed by others
REFERENCES
Tiwari, G., Tiwari, R., Bannerjee, S.K., Bhati, L., Pandey, S., Pandey, P., Sriwastawa, B., Int. J. Pharm. Investig., 2012, vol. 2, no. 1, p. 2. https://doi.org/10.4103/2230-973X.96920
Liu, D., Yang, F., Xiong, F., and Gu, N., Theranostics, 2016, vol. 6, no. 9, p. 1306. https://doi.org/10.7150/thno.14858
Sung, Y.K. and Kim, S.W., Biomater. Res., 2020, vol. 24, no. 1, p. 12. https://doi.org/10.1186/s40824-020-00190-7
Dang, Y. and Guan, J., Smart Mater. Med., 2020, vol. 1, p. 10. https://doi.org/10.1016/j.smaim.2020.04.001
Yao, Y., Zhou, Y., Liu, L., Xu, Y., Chen, Q., Wang, Y., Wu, S., Deng, Y., Zhang, J., and Shao, A., Front. Mol. Biosci., 2020, vol. 7, p. 193. https://doi.org/10.3389/fmolb.2020.00193
Amreddy, N., A. Babu, Muralidharan, R., Panneerselvam, J., Srivastava, A., Ahmed, R., Mehta, M., Munshi, A., and Ramesh, R., Adv. Cancer Res., 2018, p. 115. https://doi.org/10.1016/bs.acr.2017.11.003
Hao, M., Chen, B., Zhao, X., Zhao, N., and Xu, F.J., Mater. Chem. Front., 2020, vol. 4, no. 9, p. 2571. https://doi.org/10.1039/D0QM00323A
Castro, R.I., Forero-Doria, O., and Guzmán, L., An. Acad. Bras. Ciênc., 2018, vol. 90, no. 2, p. 2331. https://doi.org/10.1590/0001-3765201820170387
Mukherjee, S., Mukherjee, S., Abourehab, M.A.S., Sahebkar, A., and Kesharwani, P., Eur. Polym. J., 2022, vol. 177, p. 111471. https://doi.org/10.1016/j.eurpolymj.2022.111471
Kesharwani, P., Jain, K., and Jain, N.K., Prog. Polym. Sci., 2014, vol. 39, no. 2, p. 268. https://doi.org/10.1016/j.progpolymsci.2013.07.005
Hsu, H.J., Bugno, J., Lee, S., and Hong, S., WIREs Nanomed. Nanobiotechnol., 2016, vol. 9, no. 1, p. e1409. https://doi.org/10.1002/wnan.1409
Fant, K., Esbjörner, E.K., Jenkins, A., Grossel, M.C., Lincoln, P., and Nordén, B., Mol. Pharm., 2010, vol. 7, no. 5, p. 1734. https://doi.org/10.1021/mp1001312
Duncan, R. and Izzo, L., Adv. Drug Deliv. Rev., 2005, vol. 57, no. 15, p. 2215. https://doi.org/10.1016/j.addr.2005.09.019
Bodewein, L., Schmelter, F., Di Fiore, S., Hollert, H., Fischer, R., and Fenske, M., Toxicol. Appl. Pharmacol., 2016, vol. 305, p. 83. https://doi.org/10.1016/j.taap.2016.06.008
Mostovaya, O., Padnya, P., Shiabiev, I., Mukhametzyanov, T., and Stoikov, I., Int. J. Mol. Sci., 2021, vol. 22, no. 21, p. 11901. https://doi.org/10.3390/ijms222111901
Zhou, L., Russell, D.H., Zhao, M., and Crooks, R.M., Macromolecules, 2001, vol. 34, no. 11, p. 3567. https://doi.org/10.1021/ma001782j
Subbi, J., Aguraiuja, R., Tanner, R., Allikmaa, V., and Lopp, M., Eur. Polym. J., 2005, vol. 41, no. 11, p. 2552. https://doi.org/10.1016/j.eurpolymj.2005.05.031
Padnya, P.L., Andreyko, E.A., Gorbatova, P.A., Parfenov, V.V., Rizvanov, I.K., and Stoikov, I.I., RSC Adv., 2017, vol. 7, no. 3, p. 1671. https://doi.org/10.1039/C6RA24734B
Padnya, P.L., Terenteva, O.S., Akhmedov, A.A., Iksanova, A.G., Shtyrlin, N.V., Nikitina, E.V., Krylova, E.S., Shtyrlin, Y.G., and Stoikov, I.I., Bioorg. Med. Chem., 2021, vol. 29, p. 115905. https://doi.org/10.1016/j.bmc.2020.115905
Shurpik, D.N., Padnya, P.L., Stoikov, I.I., and Cragg, P.J., Molecules, 2020, vol. 25, no. 21, p. 5145. https://doi.org/10.3390/molecules25215145
Funding
This study was financially supported by the Russian Science Foundation (grant no. 21-73-20067). Registration of the mass spectra was supported by the Program for Strategic Academic Leadership of Kazan (Volga Region) Federal University (“Prioritet-2030”).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
To the 145th anniversary of A.E. Arbuzov
Supplementary information
Rights and permissions
About this article
Cite this article
Shiabiev, I.E., Pysin, D.A., Padnya, P.L. et al. First-Generation Dendrimers Based on Thiacalix[4]arene Containing Hydroxyl Terminal Groups: Synthesis and Self-Assembly. Russ J Gen Chem 92, 2574–2581 (2022). https://doi.org/10.1134/S1070363222120040
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222120040