Abstract
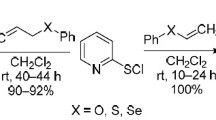
Annulation reactions of pyridine-2-sulfenyl bromide with vinyl and allyl ethers proceeded regioselectively, but with opposite regiochemistry. Reactions with vinyl ethers led to 3-organyloxy-2,3-dihydro[1,3]thiazolo[3,2-a]pyridin-4-ium bromides (91–99% yields), while allyl ethers gave 2-substituted derivatives of 2,3-dihydro[1,3]thiazolo[3,2-a]pyridin-4-ium (90–98% yields). Tricyclic condensed compounds were synthesized by reactions of pyridine-2-sulfenyl bromide with cyclic ethers (2,3-dihydrofuran, 3,4-dihydro-2H-pyran and 2,5-dihydrofuran).





Similar content being viewed by others
REFERENCES
Sharma, P.K., Amin, A., and Kumar, M., Open Med. Chem. J., 2020, vol. 14, p. 49. https://doi.org/10.2174/1874104502014010049
Chhabria, M.T., Patel, S., Modi, P., and Brahmkshatriya, P.S., Curr. Top. Med. Chem., 2016, vol. 16, p. 2841. https://doi.org/10.2174/1568026616666160506130731
Litvinov, V.P., Dotsenko, V.V., and Krivokolysko, S.G., Khimiya tienopiridinov i rodstvennykh sistem (Chemistry of thienopyridines and related systems), Moscow: Nauka, 2006, p. 174.
Nagaraju, A., Ramulu, B.J., Shukla, G., Srivastava, A., Verma, G.K., Raghuanshi, K., and Singh, M.S., Tetrahedron, 2015, vol. 71, p. 3422. https://doi.org/10.1016/j.tet.2015.03.087
Mahmoud, N.F.H. and Balamon, M.G., J. Heterocycl. Chem., 2020, vol. 57, p. 3056. https://doi.org/10.1002/jhet.4011
Boominathan, M., Nagaraj, M., Maheshwaran, C., Muthusubramanian, S., and Bhuvanesh, N., J. Heterocycl. Chem., 2014, vol. 51, p. 244. https://doi.org/10.1002/jhet.1650
El-Emary, T.I., Khalil, A., El-Hag Ali, G.A.M., and El-Adasy, A.A.A.M., Phosphorus, Sulfur, Silicon, Relat. Elem., 2005, vol. 180, p. 19. https://doi.org/10.1080/10426500490494778
Walker, K.A., Sjogren, E.B., and Matthews, T.R., J. Med. Chem., 1985, vol. 28, p. 1673. https://doi.org/10.1021/jm00149a023
Shi, F., Li, C., Xia, M., Miao, K., Zhao, Y., Tu, S., Zheng, W., Zhang, G., and Ma, N., Bioorg. Med. Chem. Lett., 2009, vol. 19, p. 5565. https://doi.org/10.1016/j.bmcl.2009.08.046
Park, H., Hwang, K.Y., Oh, K.H., Kim, Y.H., Lee, J.Y., and Kim, K., Bioorg. Med. Chem., 2008, vol. 16, p. 284. https://doi.org/10.1016/j.bmc.2007.09.036
Borisov, A.V., Osmanov, V.K., Borisova, G.N., Matsulevich, Zh.V., and Fukin, G.K., Mendeleev Commun., 2009, vol. 19, p. 49. https://doi.org/10.1016/j.mencom.2009.01.020
Potapov, V.A., Musalova, M.V., Ishigeev, R.S., Musalov, M.V., Panov, V.A., Khabibulina, A.G., Amosova, S.V., and Bhasin, K.K., Tetrahedron Lett., 2016, vol. 57, p. 5341. https://doi.org/10.1016/j.tetlet.2016.10.066
Potapov, V.A., Ishigeev, R.S., and Amosova, S.V., Russ. J. Org. Chem., 2016, vol. 52, no. 6, p. 918. https://doi.org/10.1134/S1070428016060300
Potapov, V.A., Ishigeev, R.S., Amosova, S.V., and Borodina, T.N., Tetrahedron Lett., 2019, vol. 60, p. 475. https://doi.org/10.1016/j.tetlet.2019.01.001
Potapov, V.A., Ishigeev, R.S., Shkurchenko, I.V., Zinchenko, S.V., and Amosova, S.V., Molecules, 2020, vol. 25, p. 376. https://doi.org/10.3390/molecules25020376
Potapov, V.A., Ishigeev, R.S., Shkurchenko, I.V., and Amosova, S.V., Russ. J. Gen. Chem., 2019, vol. 89, no. 12, p. 2601. https://doi.org/10.1134/S1070363219120429
Reutov, O.A., Kurts, A.L., and Butin K.P., Organicheskaya khimiya (Organic Chemistry), Moscow: Binom, Laboratoriya Znanii, 2005, vol. 1, p. 407.
Smit, V.A., Zefirov, N.S., Bodrikov, I.V., and Krimer, M.Z., Acc. Chem. Res., 1979, vol. 12, p. 282. https://doi.org/10.1021/ar50140a003
Rasteikiene, L., Greiciute, D., Lin’kova, M.G., and Knunyants, I.L., Russ. Chem. Rev., 1977, vol. 46, p. 548. https://doi.org/10.1070/RC1977v046n06ABEH002155
Abu-yousef, I.A. and Harpp, D.N., Sulfur Rep., 2003, vol. 24, p. 255. https://doi.org/10.1080/01961770308047977
Denmark, S.E. and Vogler, T., Chem. Eur. J., 2009, vol. 15, p. 11737. https://doi.org/10.1002/chem.200901377
Klimova, V.A., Osnovnye mikrometody analiza organicheskikh soedinenii (Basic Micromethods for the Analysis of Organic Compounds), Moscow: Khimiya, 1975, p. 101.
ACKNOWLEDGMENTS
Spectral measurements were carried out using the equipment of the Baikal Analytical Center for Collective Use of the Siberian Branch of the Russian Academy of Sciences.
Funding
This work was financially supported by the Russian Science Foundation (project no. 22-13-00339).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ishigeev, R.S., Amosova, S.V. & Potapov, V.A. Regioselective Reactions of Pyridine-2-sulfenyl Bromide with Vinyl and Allyl Ethers. Russ J Gen Chem 92, 1720–1727 (2022). https://doi.org/10.1134/S1070363222090146
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222090146