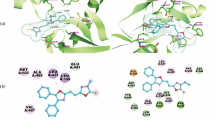
Abstract
In the study, two series of thiazolidine-2,4-dione derivatives have been designed, synthesized and evaluated for their inhibitory activity of Mcl-1 protein by fluorescence polarization assays (FPAs). Among those, the most potent product has demonstrated inhibitory activity against Mcl-1 (inhibitory rate 94% at 10 μM) higher than that of the positive control WL-276 (90%).



Similar content being viewed by others
REFERENCES
Danial, N.N., and Korsmeyer, S.J., Cell, 2004, vol. 116, p. 205. https://doi.org/10.1016/s0092-8674(04)00046-7
Negi, A., and Murphy, P.V., Eur. J. Med. Chem., 2021, vol. 210, p. 113038. https://doi.org/10.1016/j.ejmech.2020.113038
Friberg, A., Vigil, D., Zhao, B., Daniels, R.N., Burke, J.P., Garcia-Barrantes, P.M., Camper, D., Chauder, B.A., Lee, T., Olejniczak, E.T., and Fesik, S.W., J. Med. Chem., 2013, vol. 56, p. 15. https://doi.org/10.1021/jm301448p
Wan, Y.C., Dai, N.N., Tang, Z.L., and Fang, H., Eur. J. Med. Chem., 2018, vol. 146, p. 471. https://doi.org/10.1016/j.ejmech.2018.01.076
Wang, L.Y., Sloper, D.T., Addo, S.N., Tian, D., Slaton, J.W., and Xing, C.G., Cancer Res., 2008, vol. 68, p. 4377. https://doi.org/10.1158/0008-5472.CAN-07-6590
Wan, Y.C., Wu, S.L., Xiao, G.Z., Liu, T.T., Hou, X.B., Chen, C.C., Guan, P., Yang, X.Y., and Fang, H., Bioorg. Med. Chem., 2015, vol. 23, p. 1994. https://doi.org/10.1016/j.bmc.2015.03.024
Mendgen, T., Steuer, C., and Klein, C.D., J. Med. Chem., 2012, vol. 55, p. 743. https://doi.org/10.1021/jm201243p
Denis, C., Sopková-de Oliveira Santos, J., Bureau, R., and Voisin-Chiret, A.S., J. Med. Chem., 2020, 63, no. 3, p. 928. https://doi.org/10.1021/acs.jmedchem.9b00983
Wan, Y.C., Li, Y.H., Yan, C.X., Wen, J.J., and Tang, Z.L. Bioorg. Chem., 2020, vol. 104, p. 104217. https://doi.org/10.1016/j.bioorg.2020.104217
Maccari, R., Ottanà, R., Curinga, C., Vigorita, M.G., Rakowitz, D., Steindl, T., and Langer, T., Bioorg. Med. Chem., 2005, vol. 13, p. 2809. https://doi.org/10.1016/j.bmc.2005.02.026
Wan, Y.C., Wang, J.H., Sun, F.E., Chen, M.L., Hou, X.B., and Fang, H., Bioorg. Med. Chem., 2015, vol. 23, p. 7685. https://doi.org/10.1016/j.bmc.2015.11.014
Funding
This work was supported by National Natural Science Foundation of China (Grant no. 82003602, 21877034), the science and technology innovation Program of Hunan Province (Grant no. 2021RC5028), Scientific Research Fund of Hunan Provincial Education Department (Grant no. 21B0478) and Open Project Program of Key Laboratory of Theoretical Organic Chemistry and Functional Molecule, Ministry of Education (Grant no. LKF202105).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interests was declared by the authors.
Rights and permissions
About this article
Cite this article
Deng, X., Long, J., Wang, W. et al. Design, Synthesis, Biological Evaluation, and Molecular Docking Studies of Thiazolidine-2,4-dione Derivatives as Mcl-1 Inhibitors. Russ J Gen Chem 92, 464–469 (2022). https://doi.org/10.1134/S1070363222030148
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222030148