Abstract
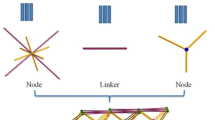
A novel two-dimensional Zn(II) coordination polymer [Zn(II) CP], [{Zn(L)Zn(OAc–)(µ-OAc–)}4]n· 2nCH3CH2OH has been self-assembled from Zn(OAc)2·2H2O. It contains pyridine-terminal salamo-type ligand H2L. Structure of the product has been determined. The Zn(II)CP contains two kinds of Zn(II) ions of different coordination modes. The Zn1 center is characterized by five-coordinated triangular biconical geometry. The Zn2 center is a six-coordinated octahedral center. Using these as junction points and the fully deprotonated ligand (L)2– moieties containing pyridine-terminal as a linker, the 2-D coordination polymer with two kinds of different pores [27.47 × 11.77(23) Å2; 11.97 × 4.51(17) Å2] has been obtained. The Zn(II) CP can fluoresce green and be used in fluorescent materials. Short-range interactions on Zn(II) CP surface have been studied by Hirshfeld surface analysis.







Similar content being viewed by others
REFERENCES
Schäfer, T., Sedykh, A.E., Becker, J., and Buschbaum, K.M., Z. Anorg. Allg. Chem., 2020, vol. 646, p. 1. https://doi.org/10.1002/zaac.202000103
Duan, J.G., Li, Y., Pan, Y., Behera, N., and Jin, W., Coord. Chem. Rev., 2019, vol. 395, p. 25. https://doi.org/10.1016/j.ccr.2019.05.018
Gao, G.P., Zheng, F., and Wang, L.W., Chem. Mater., 2020, vol. 32, p. 1974. https://doi.org/10.1021/acs.chemmater.9b04852
Liu, J.J., Lu, Y.W., Li, J., and Lu, W.B., Dyes Pigm., 2020, vol. 177, p. 108266. https://doi.org/10.1016/j.dyepig.2020.108266
Ma, N., Lin, C., Wu, N., Liu, Q., Ma, J.L., Meng, W., Wang, X.S., Zhang, L., Xu, X.H., Zhao, Y.F., Zhuang, L., Fan, J., Sun, J.L., Zhuo, R.X., and Zhang, X.Z., J. Mater. Chem. A, 2017, vol. 5, p. 23440. https://doi.org/10.1039/c7ta08002f
Stephanie, A.B. and David, R.T., Cryst. Growth Des., 2016, vol. 16, p. 6294. https://doi.org/10.1021/acs.cgd.6b00901
Wang, L.L., Wang, J.M., Fan, C.B., Bi, C.B., Zhang, X.D., Zhang, M., Wang, M., and Fan, Y.H., Appl Organomet Chem., 2020, p. e5767. https://doi.org/10.1002/aoc.5767
Nakamura, T., Tsukuda, S., and Nabeshima, T., J. Am. Chem. Soc., 2019, vol. 141, p. 6462. https://doi.org/10.1021/jacs.9b00171
Ryu, C.H., Kwak, S.W., Lee, H., Lee, H.W., Hwang, J.H., Kim, H.M., Chung, Y., Kim, Y.M., Park, M.H., and Lee, K.M., Inorg. Chem., 2019, vol. 58, p. 12358. https://doi.org/10.1021/acs.inorgchem.9b01948
Liu, C., An, X.X., Cui, Y.F., Xie, K.F., and Dong, W.K., Appl. Organomet. Chem., 2020, vol. 34, p. e5272. https://doi.org/10.1002/aoc.5272
Zhang, Y., Yu, M., Pan, Y.Q., Zhang, Y., Xu, L., and Dong, X.Y., Appl. Organomet. Chem., 2020, vol. 34, p. e5442. https://doi.org/10.1002/aoc.5442
Wang, J.F., Bian, R.N., Feng, T., Xie, K.F., Wang, L., and Ding, Y.J., Microchem. J., 2021, vol. 160, p. 105676. https://doi.org/10.1016/j.microc.2020.105676
Madison, W.I., SAINT-Plus, Bruker Analytical X-ray System; Bruker: Billerica, MA, USA, 1999.
Sheldrick, G.M., SADABS, Program for Empirical Absorption Correction of Area Detector Data; University of Gottingen: Gottingen, Germany, 1996.
Wang, J.F., Xu, X., Bian, R.N., Dong, W.K., and Ding, Y.J., Inorg. Chim. Acta, 2021, vol. 516, p. 120095. https://doi.org/10.1016/j.ica.2020.120095
Majumder, A., Rosair, G.M., Mallick, A., Chattopadhyay, N., and Mitra, S., Polyhedron, 2006, vol. 25, p. 1753. https://doi.org/10.1016/j.poly.2005.11.029
Akine, S., Morita, Utsuno, Y.F., and Nabeshima, T., Inorg. Chem., 2009, vol. 48, p. 10670. https://doi.org/10.1021/ic901372k
Dong, X.Y., Gao, L., Wang, F., Zhang, Y., and Dong, W.K., Chin. J. Inorg. Chem., 2018, vol. 34, p. 739. https://doi.org/10.11862/CJIC.2018.151
Zerdane, S., Wilbraham, L., Cammarata, M., Iasco, O., Rivière, E., Boillot, M.L., Ciofini, I., and Colle, E., Chem. Sci., 2017, vol. 8, p. 4978. https://doi.org/10.1039/C6SC05624E
Yang, L., Douglas, R.P., and Robert, P.H., Dalton Trans., 2007, vol. 7, p. 955. https://doi.org/10.1039/B617136B
Rohl, L.A., Moret, M., Kaminsky, W., Claborn, K., McKinnon, J.J., and Kahr, B., Cryst. Growth Des., 2015, vol. 8, p. 4517. https://doi.org/10.1021/cg8005212
Saikat, K.S., Acta Crystallogr. E, 2018, vol. 74, p. 600. https://doi.org/10.1107/s2056989018003857
Suparna, T., Anowar, H., Saikat, K.S., and Subrata, M., J. Mol. Struct., 2020, vol. 1216, p. 128207. https://doi.org/10.1016/j.molstruc.2020.128207
Funding
This work was supported by the National Natural Science Foundation of China (21761018), which are gratefully acknowledged. Computations were done using National Supercomputing Center in Shenzhen, P. R. China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Li, LL., Feng, SS., Zhang, JQ. et al. Coordination Properties and Fluorescence of a Structurally Novel Zn(II) Pyridine-Terminal Salamo-Type Coordination Polymer. Russ J Gen Chem 91, 1128–1134 (2021). https://doi.org/10.1134/S1070363221060220
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363221060220