Abstract
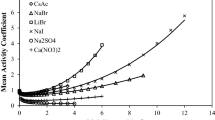
A model of a solution of non-electrolytes has been considered, which is a superposition of equilibrium processes of solvation, homo- and heteroassociation. The chemical potentials and activity coefficients of the components have been calculated for a binary solution considering only symmetric solvation. The model has been verified to calculate the density of binary solutions with high agreement with experimental data.
Similar content being viewed by others
References
Smirnova, N.A., Molekulyarnye teorii rastvorov (Molecular Theory of Solutions), Leningrad: Khimiya, 1987.
Prigizhin, I.P., Molekulyarnaya teoriya rastvorov (Molecular Theory of Solutions), Moscow: Metallurgiya, 1990.
Terziyan, T.V., Safronov, A.P., and Sabirova, A.R., Russ. J. Phys. Chem. (A), 2017, vol. 91, no. 2, p. 1229. https://doi.org/10.1134/S0036024417070299
Balankina, E.S., Russ. J. Phys. Chem. (A), 2016, vol. 90, no. 6, p. 1157. https://doi.org/10.1134/S0036024416050095
Timofeev, M.V., Zh. Fiz. Khim., 2014, vol. 88, no. 9, p. 1354.
Zevatskii, Yu.E., Sb. tezisov IX nauchno-tekhnicheskoi konf. studentov, aspirantov i molodykh uchenykh v ramkakh meropriyatii, posvyashchennykh 150-letiyu otkrytiya Periodicheskogo zakona khimicheskikh elementov D.I. Mendeleevym “Nedelya nauki-2019” (Proc. of IX Sci. and Techn. Conf. of Students, Graduate Students, and Young Scientists in the Framework of Events Dedicated to the 150th Anniversary of the Discovery of the Periodic Law of Chemical Elements Mendeleev “Science Week — 2019”), St. Petersburg: SPbGTI(TU), 2019, p. 4.
Teoreticheskie i eksperimental’nye metody khimii rastvorov (Problemy khimii rastvorov) [Theoretical and Experimental Methods of Chemistry of Solutions (Problems of Chemistry of Solutions)], Tsivadze, A.Yu., Ed., Moscow: Prospekt, 2011.
Walas, S.M., Phase Equilibria in Chemical Engineering, Butterworths: Stoneham, MA, 1984.
Van Laar, J.J., Z. Phys. Chem., 1910, vol. 72, p. 723. https://doi.org/10.1515/zpch-1910-7236
Wilson, G.M., J.Am. Chem. Soc., 1964, vol. 86, no. 2, p. 127. https://doi.org/10.1021/ja01056a002
Renon, H., Asselineau, L., Cohen, G., and Raimbault, C., Calcul sur Ordinateur des Equilibres Liquide-Vapeur et Liquide-Liquide, Paris: Ed. Technip, 1971.
Zevatskii, Yu.E., Doctoral (Chem.) Dissertation, St. Petersburg, 2010.
Porshnev, S.V. and Belenkova, I.V., Chislennye metody na baze Mathcad (Mathcad-Based Numerical Methods), St. Petersburg: BKhV-Peterburg, 2005.
Krestov, G.A., Afanas’ev, V.N., and Efremova, L.S., Fiziko-khimicheskie svoistva binarnykh rastvoritelei (Physico-Chemical Properties of Binary Solvents), Leningrad: Khimiya, 1988.
Novyi spravochnik khimika i tekhnologa. Khimicheskoe ravnovesie. Svoistva rastvorov (New Reference Chemist and Technologist. Chemical Equilibrium. Solution Properties), St. Petersburg: NPO “Professional,” 2007.
Fedyaeva, O.A. and Poshelyuzhnaya, E.G., Russ. J. Phys. Chem. (A), 2017, vol. 91, no. 1, p. 63. https://doi.org/10.1134/S003602441701006X
Acknowledgments
The author acknowledges V.V. Gusarov for valuable comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
No conflict of interest was declared by the authors.
Russian Text © The Author(s), 2019, published in Zhurnal Obshchei Khimii, 2019, Vol. 89, No. 11, pp. 1724–1731.
Rights and permissions
About this article
Cite this article
Zevatsky, Y.E. Model of Solvation and Association Processes in Non-Electrolytes Solutions. Russ J Gen Chem 89, 2239–2245 (2019). https://doi.org/10.1134/S1070363219110136
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219110136