Abstract
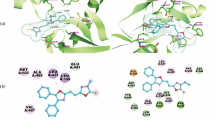
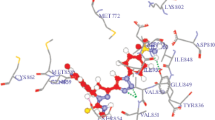
A novel series of thiophene-2-carboxamide derivatives are designed and synthesized, and their structures are confirmed by 1H and 13C NMR, and mass spectra. The synthesized compounds are evaluated for their in vitro cytotoxic activity by MTT assay. Among the tested compounds, the derivative with 4-Cl-phenyl ring exhibits potent inhibitory activity against MCF-7, K562, HepG2, and MDA-MB-231. The molecular docking study performed for the synthesized compounds against PTP1B exhibits essential key interactions.
Similar content being viewed by others
References
Basu, S., Prasad, U.V., Barawkar, D.A., De, S., Palle, V.P., Menon, S., Patel, M., Thorat, S., Singh, U.P., Sarma, K.D., Waman, Y., Niranjan, S., Pathade, V., Gaur, A., Reddy, S., and Ansari, S., Bioorg. Med. Chem. Lett., 2012, vol. 22, p. 2843. doi https://doi.org/10.1016/j.bmcl.2012.02.070
Gulipalli, K.C., Bodige, S., Ravula, P., Endoori, S., Vanaja, G.R., Suresh, B.G., Narendra Sharath Chandra, J.N., and Seelam, N., Bioorg. Med. Chem. Lett., 2017, vol. 27, p. 3558. doi https://doi.org/10.1016/j.bmcl.2017.05.047
Hirth, B.H., Qiao, S., Cuff, L.M., Cochran, B.M., Pregel, M.J., Gregory, J.S., Sneddon, S.F., and Kane, Jr., J.L., Bioorg. Med. Chem. Lett., 2005, vol. 15, p. 2087. doi https://doi.org/10.1016/j.bmcl.2005.02.041
Chohan, Z.H., Farooq, M.A., Scozzafava, A., and Supuran, C.T., J. Enzyme Inhib. Med. Chem., 2002, vol. 17, p. 1. doi https://doi.org/10.1080/14756360290005598
Hosseini, Z., Akbari, N., Gholivand, K., Farshadian, S., and Khajeh, K., Appl. Organomet. Chem., 2010, vol. 24, p. 700. doi https://doi.org/10.1002/aoc.1669
Mahmood, N., Jhaumeer-Lauloo, S., Sampson, J., and Houghton, P.J., J. Pharm. Pharmacol., 1998, vol. 50, p. 1339. doi https://doi.org/10.1111/j.2042-7158.1998.tb03356.x
Pejchalová, M., Havelek, R., Královec, K., Růžičková, Z., and Pejchal, V., Med. Chem. Res., 2017, vol. 26, p. 1847. doi https://doi.org/10.1007/s00044-017-1894-x
Fun, H.K., Pai, K.S.R., Shetty, P., Isloor, A.M., and Sankappa, R.U., Arab. J. Chem., 2014, vol. 8, p. 317. doi https://doi.org/10.1016/j.arabjc.2014.01.018
Li, W.S., More, S.V., Wang, C.H., Jen, Y.C., Yao, C.F., Wang, T.F., Hung, C.C., and Jao, S.C., Bioorg. Med. Chem. Lett., 2010, vol. 20, p. 1148. doi https://doi.org/10.1016/j.bmcl.2009.12.017
Mou, J., Fang, H., Jing, F., Wang, Q., Liu, Y., Zhu, H., Shang, L., Wang, X., and Xu, W., Bioorg. Med. Chem., 2009, vol. 17, p. 4666. doi https://doi.org/10.1016/j.bmc.2009.04.056
Bodige, S., Ravula, P., Gulipalli, K.C., Endoori, S., Narendra Sharath Chandra, J.N., Cherukumalli, P.K.R., Vanaja, G.R., and Seelam, N., Anticancer Agents Med. Chem., 2018, vol. 18, p. 891. doi https://doi.org/10.2174/1871520618666180209151018
Ravula, P., Vamaraju, H.B., Paturi, M., Bodige, S., Gulipalli, K.C., and Narendra Sharath Chandra, J.N., J. Heterocycl. Chem., 2018, vol. 55, p. 1313. doi https://doi.org/10.1002/jhet.3163
Ye, D., Zhang, Y., Wang, F., Zheng, M., Zhang, X., Luo, X., Shen, X., Jiang, H., and Liu, H., Bioorg. Med. Chem., 2010, vol. 18, p. 1773. doi https://doi.org/10.1016/j.bmc.2010.01.055
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gulipalli, K.C., Ravula, P., Bodige, S. et al. Synthesis and Anticancer Activity of Thiophene-2-carboxamide Derivatives and In Silico Docking Studies. Russ J Gen Chem 89, 1502–1512 (2019). https://doi.org/10.1134/S1070363219070211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219070211