Abstract
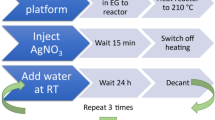
General regularities of nanoparticles formation from solutions under various conditions (organic and aqueous media, mild and rigid conditions) are presented and analyzed. Two ways of a a solid phase formation, based on simultaneous or sequential crystallization, are proposed; examples of the corresponding synthetic procedures are given. The process of sequential crystallization is described. The ability to reduce or increase particle sizes by using morphology control agents has been demonstrated by specific examples. Nucleating agents or nuclei can be successfully used to reduce the size of particles or to obtain the desired crystalline phases (if the compound is characterized by several polymorphs).
Similar content being viewed by others
References
Suslonov, V.V., Martines-Rodriges, N., Mingabudi-nova, L.R., Osmolovskii, M.G., and Osmolovskaya, O.M., Russ. J. Gen. Chem., 2015, vol. 85, no. 7, p. 1768. doi https://doi.org/10.1134/s1070363211110260
Fedotov, M.A., Krivoruchko, O.P., and Buyanov, R.A., Izv. Akad. Nauk SSSR, Ser. Khim, 1977, no. 10, p. 2183.
Osmolovskii, M.G., Osmolovskaya, O.M., and Gordeev, S.V., Vestn. Nat. Res. Nucl. Univ., 2012, vol. 1, no. 1, p. 80.
Matveeva, P.A., Nazarov, D.V., Osmolovskaya, O.M., Kasatkin, Yu.A., Smirnov, V.M., Bobrysheva, N.P., and Osmolovskii, M.G., Russ. J. Gen. Chem., 2015, vol. 85, no. 1, p. 208. doi https://doi.org/10.1134/S1070363215010387
Vakh, C., Pochivalov, A., Podurets, A., Bobrysheva, N., Osmolovskaya, O., and Bulatov A., Analyst, 2019, vol. 144, no. 1, p. 148. doi https://doi.org/10.1039/C8AN01868E
Kotel’nikova, S.V., Suslonov, V.V., Bobrysheva, N.P., Osmolowsky, M.G., and Osmolowskaya, O.M., Russ. J. Gen. Chem. 2017, vol. 87, no. 5, p. 1093. doi https://doi.org/10.1134/s1070363217050334
Timofeeva, I., Alikina, M., Vlasova, A., Osmolowsky, M., Voznesenskiy, M., Volina, O., Moskvin, L., Osmolovskaya, O., and Bulatov, A., Analyst, 2019, vol. 144, p. 152. doi https://doi.org/10.1039/c8an01894d
Suslonov, V.V., Osmolovskaya, O.M., and Osmolowsky, M.G., Russ. J. Gen. Chem., 2012, vol. 82, no. 9, p. 1585. doi https://doi.org/10.1134/s107036321209023x
Kotelnikova, S.V., Suslonov, V.V., Voznesenskiy, M.A., Bobrysheva, N.P., Osmolowsky, M.G., Rajabi, F., and Osmolovskaya, O.M., Mater. Chem. Phys., 2019, vol. 223, p. 745. doi https://doi.org/10.1016/j.matchemphys.2018.11.046
Osmolowsky, M.G. and Osmolovskaya, O.M., Bull. Russ. Acad. Sci. Phys., 2012, vol. 76, no. 10, p. 1146. doi https://doi.org/10.3103/S1062873812100085
Petukhova, Y.V., Osmolowskaya, O.M., and Osmolowsky, M.G., J. Phys. Conf. Ser., 2015, vol. 643, p. 012121. doi https://doi.org/10.1088/1742-6596/643/1/012121
Osmolowskaya, O.M., Regulation of HAP and Iron Oxide Nanoparticle Morphology Using Chelating Agents. Biogenic-Abiogenic Interactions in Natural and Anthropogenic Systems. Lecture Notes in Earth System Sciences, Switzerland: Springer International Publishing, 2016, p. 543. doi https://doi.org/10.1007/978-3-319-24987-2_36
Kozlova, M.A., Poezzhaev, A.I., Khramenkova, E.V., Korzhikov, V.A., Osmolovskaya, O.M., and Osmolowsky, M.G., Russ. J. Gen. Chem., 2014, vol. 84, no. 10, p. 2047. doi https://doi.org/10.1134/s1070363214100338
Arkhipov, D.I., Bobrysheva, N.P., Dzidziguri, E.L., Osmolowsky, M.G., and Osmolovskaya, O.M., J. Therm. Anal. Calorim., 2017, vol. 128, no. 1, p. 71. doi https://doi.org/10.1007/s10973-016-5919-3
Arkhipov, D.I., Gordeev, S.V., Dzidziguri, E.L., Osmolowsky, M.G., Osmolovskaya, O.M., Russ. J. Gen. Chem., 2016, vol. 86, no. 4, p. 983. doi https://doi.org/10.1134/S107036321604037X
Sarnovskii-González, A.D., Kudinova, A.A., Petukhova, Y.V., Bobrysheva, N.P., Osmolowsky, M.G., and Osmolovskaya, O.M., Russ. J. Gen. Chem., 2018, vol. 88, no. 10, p. 2240. doi https://doi.org/10.1134/s1070363218100390
Petukhova, Y.V., Mosiagin, I.P., Mezenov, I.A., Sarnovskiy-Gonzalez, A.D., Ubyivovk, E.V., Bobrysheva, N.P., and Osmolovskaya, O.M., J. Phys. Chem. Solids, 2018, vol. 121, p. 128. doi https://doi.org/10.1016/j.jpcs.2018.05.002
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Obshchei Khimii, 2019, Vol. 89, No. 6, pp. 937–945.
Funding
This work was financially supported by the Russian Foundation for Basic Research (grants nos. 18-03-01066-a, 16-03-00167-a, and 13-03-00943-a) with the use of equipment of the resource centers “Chemical Analysis and Materials Research Centre,” “Centre for X-ray Diffraction Studies,” “Center for Optical and Laser Materials Research,” “Centre for Physical Methods of Surface Investigation,” “Centre for Innovative Technologies of Composite Nanomaterials,” “Nanophotonics Centre,” “Magnetic Resonance Research Centre,” “Cryogen sector,” and “Chemistry Educational Centre” of St. Petersburg State University.
Conflict of Interest
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Osmolovskaya, O.M., Petukhova, Y.V., Podurets, A.A. et al. Approaches to the Control of Morphological Parameters of Inorganic Nanoparticles in the Synthesis from Solutions. Russ J Gen Chem 89, 1154–1161 (2019). https://doi.org/10.1134/S1070363219060094
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219060094