Abstract
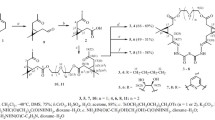
Acylation of cone and 1,3-alternate stereoisomers of hydrazides of p-tert-butylthaicalix[4]arene tetrasubstituted at the lower rim with S,S-lactide in dimethylsulfoxide has afforded the macrocyclic oligolactide derivatives containing 16 (cone) to 24 (1,3-alternate) lactide fragments. It has been shown that nanosized aggregates (80 nm) are formed with silver nitrate in the case of the cone stereoisomer containing the oligolactide fragments at one side of the macrocyclic rim, whereas submicron particles (400 nm) are formed in the case of the 1,3-alternate conformation when the substituents are at different sides of the macrocycle.
Similar content being viewed by others
References
Wan, P., Yuan, C., Tan, L., Li, Q., and Yang, K., Comp. Sci. Tech., 2014, vol. 98, p. 36. doi 10.1016/j.compscitech.2014.04.011
Knutsen, A.R., Borkowski, S.L., Ebramzadeh, E., Flanagan, C.L., Hollister, S.J., and Sangiorgio, S.N., J. Mech. Behav. Biomed. Mater., 2015, vol. 49, p. 332. doi 10.1016/j.jmbbm.2015.05.015
Comăniță, E.D., Hlihor, R.M., Ghinea, C., and Gavrilescu, M., Env. Eng. Manag. J., 2016, vol. 15, no. 3, p. 675.
Richert, A., and Olewnik-Kruszkowska, E., Ecol. Quest., 2018, vol. 29, no. 2, p. 1. doi 10.12775/EQ.2018.014
Hamad, K., Kaseem, M., Yang, H.W., Deri, F., and Ko, Y.G., Express Polym. Lett., 2015, vol. 9, no. 5, p. 435. doi 10.3144/expresspolymlett.2015.42
Timashev, P., Kuznetsova, D., Koroleva, A., Prodanets, N., Deiwick, A., Piskun, Y., and Rochev, Y., Nanomed., 2016, vol. 11, no. 9, p. 1041. doi 10.2217/nnm-2015-0022
Kang, H., Li, Y., Gong, M., Guo, Y., Guo, Z., Fang, Q., and Li, X., RSC Adv., 2018, vol. 8, no. 21, p. 11643. doi 10.1039/C7RA13448G
Brzezinski, M. and Biela, T., Macromolecules, 2015, vol. 48, no. 9, p. 2994. doi 10.1021/acs.macromol.5b00208
Gorbachuk, V.V., Mostovaya, O.A., Evtugyn, V.G., Osin, Y.N., Rizvanov, I.K., Gerasimov, A.V., and Stoikov, I.I., Macroheterocycles, 2017, vol. 10, no. 2, p. 174. doi 10.6060/mhc170513s
Gorbatchuk, V.V., Porfireva, A.V., Stepanova, V.B., Kuzin, Y.I., Evtugyn, V.G., Shamagsumova, R.V., Stoikov, I.I., and Evtugyn, G.A., Sens. Actuators (B), 2017, vol. 246, p. 136. doi 10.1016/j.snb.2017.02.061
Porifreva, A.V., Gorbatchuk, V.V., Evtugyn, V.G., Stoikov, I.I., and Evtugyn, G.A., Electroanal., 2018, vol. 30, no. 4, p. 641. doi 10.1002/elan.201700638
Andreyko, E.A., Padnya, P.L., and Stoikov, I.I., Colloids Surf. (A), 2014, vol. 454, p. 74. doi 10.1016/j.colsurfa.2014.04.021
Sýkora, J., Himl, M., Stibor, I., Císařová, I., and Lhoták, P., Tetrahedron, 2007, vol. 63, no. 10, p. 2244. doi 10.1016/j.tet.2006.12.070
Yushkova, E.A., Stoikov, I.I., Puplampu, J.B., Antipin, I.S., and Konovalov, A.I., Langmuir, 2011, vol. 27, no. 23, p. 14053. doi 10.1021/la202536x
Stoikov, I.I., Yushkova, E.A., Zharov, I., Antipin, I.S., and Konovalov, A.I., Tetrahedron, 2009, vol. 65, no. 34, p. 7109. doi 10.1016/j.tet.2009.06.045
Yakimova, L.S., Gilmanova, L.H., Evtugyn, V.G., Osin, Y.N., and Stoikov, I.I., J. Nanopart. Res., 2017, vol. 19, no. 5, p. 173. doi 10.1007/s11051-017-3868-9
Mostovaya, O.A., Padnya, P.L., Vavilova, A.A., Shurpik, D.N., Khairutdinov, B.I., Mukhametzyanov, T.A., and Stoikov, I.I., New J. Chem., 2018, vol. 42, no. 1, p. 177. doi 10.1039/C7NJ03953K
Luckachan, G.E. and Pillai, C.K.S., Carbohydr. Polym., 2006, vol. 64, no. 2, p. 254. doi 10.1016/j.carbpol.2005.11.035
Bahloul, M., Chamignon, C., Pruvost, S., Fleury, E., Charlot, A., and Portinha, D., Polymer, 2015, vol. 79, p. 195. doi 10.1016/j.polymer.2015.10.025
Kumar, R., Lee, Y.O., Bhalla, V., Kumar, M., and Kim, J.S., Chem. Soc. Rev., 2014, vol. 43, no. 13, p. 4824. doi 10.1039/c4cs00068d
Stoikov, I.I., Yantemirova, A.A., Nosov, R.V., Rizvanov, I.Kh., Julmetov, A.R., Klochkov, V.V., Antipin, I.S., Konovalov, A.I., and Zharov, I., Org. Biomol. Chem., 2011, vol. 9, no. 9, p. 3225. doi 10.1039/c0ob01251c
Galukhin, A.V., Shabalin, K.V., Antipin, I.S., Konovalov, A.I., and Stoikov, I.I., Mendeleev Commun., 2013, vol. 23, no. 1, p. 41. doi 10.1016/j.mencom.2013.01.015
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the 110th anniversary of M.I. Kabachnik’s birth
Original Russian Text © V.V. Gorbachuk, P.L. Padnya, I.I. Stoikov, 2018, published in Zhurnal Obshchei Khimii, 2018, Vol. 88, No. 9, pp. 1493–1497.
Rights and permissions
About this article
Cite this article
Gorbachuk, V.V., Padnya, P.L. & Stoikov, I.I. Synthesis of Macrocyclic Stereoisomers Substituted with Oligolactide Fragments. Russ J Gen Chem 88, 1838–1841 (2018). https://doi.org/10.1134/S1070363218090128
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363218090128