Abstract
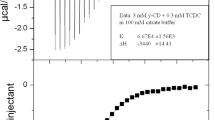
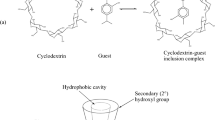
Complex formation of α-, β-, and γ-cyclodextrins with sulfasalazine in biologically relevant buffer solutions (pH 1.2 and 7.4) was studied. Formation of inclusion complexes of 1 : 1 stoichiometry between the drug and cyclodextrins was revealed. The ionization state of sulfasalazine more strongly affects the complex formation than does the structure of cyclodextrins. Compared with a neutral molecule, sulfasalazine anionic species forms with cyclodextrins more stable complexes, which are essentially stabilized by possible surface interactions.
Similar content being viewed by others
References
Badokin, V.V., Nauchno-Prakt. Revmatol., 2005, no. 4, p.47.
Clarysse, S., Brouwers, J., Tack, J., Annaert, P., and Augustijins, P., Eur. J. Pharm. Sci., 2011, vol. 43, p. 260. doi 10.1016/j.ejps.2011.04.016
Rajesh, A., Sangeeta, A., Lamba, H.S., Anil, B., and Sandeep, K., Int. Res. J. Pharm., 2011, vol. 2, p.200.
Persson, L.C., Porter, C.J.H., Charman, W.N., and Bergström, C.A.S., Pharm. Res., 2013, vol. 30, p. 3225. doi 10.1007/s11095-013-1083-7
Oprea, T.I. and Gottfries, J., J. Molec. Graph. Model., 1999, vol. 17, p. 261. doi 10.1016/S1093-3263(99)00034-0
Wang, Q., Strab, R., Kardos, P., Ferguson, C., Li, J., Owen, A., and Hidalgo, I.J., Int. J. Pharm., 2008, vol. 356, p. 12. doi 10.1016/j.ijpharm.2007.12.024
Nasonov, E.L., Ukr. Revmatol. Zh., 2002, no. 2, p.9.
Nikuhj, K., Rajesh, A., Sangeeta, A., Chirag, P., and Dhruv, M., Int. Res. J. Pharm., 2012, vol. 3, p.221.
Rajesh, A., Sangeeta, A., Lamba, H.S., Anil, B., and Sandeep, K., Res. J. Pharm. Technol., 2012, vol. 5, p.53.
Cairo, P., Ortuso, F., Alcaro, S., Fontananova, E., Tocci, E., and Drioli, E., Chem. Phys. Lett., 2008, vol. 454, p. 374. doi 10.1016/j.cplett.2008.02.050
Chaudhary, V.B. and Patel, J.K., Int. J. Pharm. Sci. Res., 2013, vol. 4, p.68.
Loftsson T. and Duchene, D., Int. J. Pharm., 2007, vol. 329, p. 1. doi 10.1016/j.ijpharm.2006.10.044
Benesi, H.A. and Hildebrand, J.H., J. Am. Chem. Soc., 1949, vol. 71, p. 2703.
Job, P., Annal. Chim., 1928, vol. 9, p.113.
Box, K., Bevan, C., Comer, J., Hill, A., Allen, R., and Reynolds, D., Anal. Chem., 2003, vol. 75, p. 883. doi 10.1021/ac020329y
Newton, D.W. and Kluza, R.B., Drug Intell. Clin. Pharm., 1978, vol. 12, p.546.
Shalaeva, M., Kenseth, J., Lombardo, F., and Bastin, A., J. Pharm. Sci., 2008, vol. 97, p. 2581.
Vasil’ev, V.P., Borodin, V.A., and Kozlovskii, E.V., Primenenie EVM v khimiko-analiticheskikh raschetakh (Application of Computers in Chemicoanalytical Calculations), Moscow: Vysshaya Shkola, 1993.
Kerns, E.H. and Di, L., Drug-Like Properties: Concepts, Structure Design, and Methods: from ADME to Toxicity Optimization, Amsterdam: Academic, 2008.
Higuchi, T. and Connons, K.A., Advances in Analytical Chemistry and Instrumentation, Reilly, C.N., Ed., New York: Wiley-Interscience, 1965, vol. 4, p. 117.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.Yu. Kuranov, E.S. Chibunova, T.V. Volkova, I.V. Terekhova, 2016, published in Rossiiskii Khimicheskii Zhurnal, 2016, Vol. 60, No. 1, pp. 55–61.
Rights and permissions
About this article
Cite this article
Kuranov, D.Y., Chibunova, E.S., Volkova, T.V. et al. Complex Formation of Cyclodextrins with Sulfasalazine in Buffer Solutions. Russ J Gen Chem 88, 1325–1330 (2018). https://doi.org/10.1134/S1070363218060439
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363218060439