Abstract
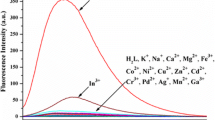
Two novel corrole-appended 18-crown-6 receptors differing in the binding manners and their aluminum complexes were designed and synthesized as potential fluoroionophores for recognition of physiologically important metal ions. In receptor 2, the corrole fluorophore was linked directly to the crown ether through an amide bond in an unbent manner, whereas in receptor 5, it was linked by a xanthene spacer in a face-to-face manner. Preliminary photophysical data show that Al-corrole complex 6 can serve as a chemosensor with selective fluorescent response towards K+ ions in CH3CN. The chemosensor showed high sensitivity owing to the change in its dual emission after binding with K+ ions. With complex 6 as fluoroionophore (5.0 × 10–6 M), the detection limit for the analysis of K+ ions was estimated to be 1.6 × 10–6 M in CH3CN.
Similar content being viewed by others
References
Kim, S.K., Lee, S.H., Lee, J.Y., Lee, J.Y., Bartsch, R.A., and Kim, J.S., J. Am. Chem. Soc., 2004, vol. 126, p. 16499.
He, H.R., Mortellaro, M.A., Leiner, M.J.P., Fraatz, R.J., and Tusa, J.K., J. Am. Chem. Soc., 2003, vol. 125, p. 1468.
Zhang, B., Ju, X.J., Xie, R., Liu, Z., Pi, S.W., and Chu, L.Y., J. Phys. Chem. B, 2012, vol. 116, p. 5527.
Miyake, M., Chen, L.D., Pozzi, G., and Bühlmann, P., Anal. Chem., 2012, vol. 84, p. 1104.
Xia, W.S., Schmehl, R.H., and Li, C.J., J. Am. Chem. Soc., 1999, vol. 121, p. 5599.
Gokel, G.W., Leevy, W.M., and Weber, M.E., Chem. Rev., 2004, vol. 104, p. 2723.
de Silva, A.P., Gunaratne, H.Q.N., Gunnlaugsson, T., Huxley, A.T.M., McCoy, C.P., Rademacher, J.T., and Rice, T.E., Chem. Rev., 1997, vol. 97, p. 1515.
Yu, H., Ju, X., Xie, R., Wang, W., Zhang, B., and Chu, L., Anal. Chem., 2013, vol. 85, p. 6477.
Gross, Z., Golubkov, G., and Simkhovich, L., Angew. Chem., Int. Ed., 2000, vol. 39, p. 4045.
Golubkov, G., Bendix, J., Gray, H.B., Mahammed, A., Goldberg, I., DiBilio, A.J., and Gross, Z., Angew. Chem., Int. Ed., 2001, vol. 40, p. 2132.
A. Mahammed, H.B., Gray, A.E., Meier-Callahan, Z., and Gross, J. Am. Chem. Soc., 2003, vol. 125, p. 1162.
Ramdhanie, B., Telser, J., Caneschi, A., Zakharov, L.N., Rheingold, A.L., and Goldberg, D.P., J. Am. Chem. Soc., 2004, vol. 126, p. 2515.
Bendix, J., Dmochowski, I.J., Gray, H.B., Mahammed, A., Simkhovich, L., and Gross, Z., Angew. Chem. Int. Ed., 2000, vol. 39, p. 4048.
Gao, Y., Liu, J., Wang, M., Na, Y., Åkermark, B., and Sun, L., Tetrahedron, 2007, vol. 63, p. 1987.
Littler, B.J., Miller, M.A., Hung, C.H., Wagner, R.W., O’Shea, D.F., Boyle, P.D., and Lindsey, J.S., J. Org. Chem. 1999, vol. 64, p. 1391.
Kowalska, D., Liu, X., Tripathy, U., Mahammed, A., Gross, Z., Hirayama, S., and Steer, R.P., Inorg. Chem., 2009, vol. 48, p. 2670.
Lazarides, T., Charalambidis, G., Vuillamy, A., Eglier, M.R., Klontzas, E., Froudakis, G., Kuhri, S., Guldi, D.M., and Coutsolelos, A.G., Inorg. Chem., 2012, vol. 51, p. 4193.
Lazarides, T., Kuhri, S., Charalambidis, G., Panda, M.K., Guldi, D.M., and Coutsolelos, A.G., Inorg. Chem., 2012, vol. 51, p. 4193.
Kita, M.R. and Miller, A.J.M., J. Am. Chem. Soc., 2014, vol. 136, p. 14519.
Shortreed, M., Kopelman, R., Kuhn, M., and Hoyland, B., Anal. Chem., 1996, vol. 68, p. 1414.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Xia, M., Sun, Xy. Selective recognition of K+ ions through change in the dual emission of a fluorescent chemosensor based on aluminum corrole-crown ether. Russ J Gen Chem 85, 2845–2852 (2015). https://doi.org/10.1134/S1070363215120348
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215120348