Abstract
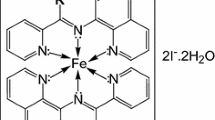
Kinetic study of some novel high spin Fe(II) complexes of Schiff base ligands derived from 5-bromsalicyaldehyde and amino acids with the OH− ion and DNA has been carried out. Based on the kinetic data, the rate law and a plausible mechanism were proposed. Kinetic data of the base catalyzed hydrolysis imply pseudo first-order double stage process due to the presence of mer- and fac-isomers. The observed rate constants k obs were correlated with the effect of a substituent R in the structure of ligands. The rate constants and activation parameters are in good agreement with stability constants of the studied complexes. Reactivity of the complexes towards DNA correlated well with the reported binding constants.
Similar content being viewed by others
References
Luo, H., Fanwick, P.E., and Green, M.A., Inorg. Chem., 1998, no 37, p. 1127
Mohamed, G.G., Omar, M.M., and Hindy, A.M.M., Spectrochim. Acta A, 2005, no 62, p. 1140.
El-Behery, M. and El-Twigry, H., Spectrochim. Acta A, 2007, no 66, p. 28.
Abdel-Rahman, L.H., El-Khatib R.M., Nassr, L.A.E., Abu-Dief, A.M., and Lashin, F.E., Spectrochim. Acta, 2013, no 111, p. 266.
Abdel-Rahman, L.H., El-Khatib, R.M., Nassr, L.A.E., Abu-Dief, A.M., Ismael, M., and Seleem, A.A., Spectrochim. Acta, 2014, no 117, p. 366.
Abbo, H.S., Titinchic, S.J.J., Prasad, R., and Chand, S., J. Mol. Catal. A: Chem., 2005, no 225, p. 225.
Gupta, K.S. and Sutar, A.K., J. Mol. Catal. A: Chem., 2007, no 272, p. 64.
Shyu, H., Wei, H., Lee, G., and Wang, Y., J. Chem. Soc., Dalton Trans., 2000, p. 915.
Wu, Z., Xu, D., and Feng, Z., Polyhedron, 2001, no 20, p. 281.
Nath, M. and Kamaluddin, S., J. Cheema, Ind. J. Chem. A, 1993, vol. 32, no 2, p. 108.
Gupta, K.S., Abdulkadir, H.K., and Chand, S., Mol. Catal. A: Chem., 2003, no 202, p. 253.
Burgess., J. and Prince, R.H., J. Chem. Soc., 1963, p. 5752.
Burgess, J., J. Chem. Soc., A., 1967, p. 431
Burgess, J., Inorg. Chim. Acta, 1971, no 5, p. 133.
Burgess, J. and Prince, R.H., J. Chem. Soc., 1965, p. 4697.
Burgess, J. and Prince, R.H., J. Chem. Soc., 1965, p. 6061.
Burgess, J. Prince, R.H., 1965) J. Chem. Soc., A), 434.
Burgess, J., J. Chem. Soc. A, 1968, p. 497.
Krumholz, P., Struct. Bonding, 1971, no 9, p. 139.
Alshehri, S., Burgess, J., and Shaker, A.M., Trans Met. Chem., 1998, no 23, p. 689.
Canali, L. and Sherrington, D.C., Chem. Soc. Rev., 1999, no 28, p. 85.
Abdel-Rahman, L.H., El-Khatib, R.M., Nassr, L.A.E., and Abu-Dief, A.M., J. Mol. Str., 2013, no 1040, p. 9.
Sharm, P.K. and Dubey, S.N., Indian J. Chem. A, 1994, vol. 33, no 12, p. 1113.
Abu-Gharib Ezz, A., EL-Khatib, R.M., Nassr, L.A.E., and Abu-Dief, A.M., Z. Phys. Chem., 2011, no 225, p. 1.
Abu-Gharib Ezz, A., EL-Khatib, R.M., Nassr, L.A.E. and Abu-Dief, A.M., J. Korean Chem. Soc., 2011, vol. 50, no 3, p. 346.
Abu-Gharib Ezz, A., EL-Khatib, R.M., Nassr, L.A.E. and Abu-Dief, A.M., Kinet. Catal., 2012, vol. 53, no 2, p. 182.
Blandamer, M.J., Burgess, J., Elvidge, D.L., Guardado, P., Hakin, A.W., Prouse, L.J.S., Radulovic, S., and Russell, D.R., Transition Met. Chem., 1991, no 16, p. 82.
Vichi, E.J.S and Kurmholz, P., J. Chem. Soc., Dalton Trans., 1975, p. 1543.
Alshehri, S., Burgess, J., and Shaker, A.M., Trans. Met. Chem., 1998, vol. 23, p. 689.
Blandamer, M.J., Burgess, J., Coohson, P., Roberts, D.L., Willings, P., Mekhail, F.M., and Askalani, P., J. Chem. Soc., Dalton Trans., 1978, p. 996.
Alshehri, S., Blandamer, M.J., Burgess, J., Guardado, P., and Hurbbard, C.D., Polyhedron, 1993, vol. 12, no 5, p. 445.
Burgess, J., Ellis, G.E., Evans, D.J., Porter, A., Wane, R., and Wyvill, R.D., J. Chem. Soc. A, 1971, p. 44.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Abdel-Rahman, L.H., El-Khatib, R.M., Nassr, L.A.E. et al. Kinetic study of the hydroxide ion attack on and DNA interaction with high spin iron(II) Schiff base amino acid chelates baering ONO donors. Russ J Gen Chem 84, 1830–1836 (2014). https://doi.org/10.1134/S1070363214090321
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363214090321