Abstract
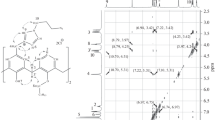
A series of disubstituted p-tert-butylcalix[4]arenes with N-methoxycarbonylmonoazacrown ether and N-ethoxymonoazacrown ether residues at the lower rim has been prepared via the reaction of di(carboxymethoxy)-p-tert-butylcalix[4]arene with azacrown ethers and subsequent reduction of the resulting amide derivatives. Using UV titration and 1H NMR spectroscopy we have demonstrated the ability of the calixarene with two N-carbonylmonoaza-18-crown-6-ether substituents to form the 1:3 complexes with K+ and Na+ and the 1:2 complexes with Cs+, Sr2+, Cu2+, and Zn2+. The calixarene with two fragments of N-ethoxymonoazo-18-crown ether has formed binuclear complexes with alkali metals cations and mononuclear complexes with transition metals cations.
Similar content being viewed by others
References
Gutsche, C.D., Calixarenes, An Introduction, 2nd ed., Cambridge: RSC Publishing, 2008.
Creaven, B.S., Donlon, D.F., and McGinley, J., Coord. Chem. Rev., 2009, vol. 253, p. 893.
Vicens, J. and Harrowfield, J., Calixarenes in the Tanoworld, Dordrecht: Springer, 2007, p. 394.
Oueslati, I., Tetrahedron, 2007, vol. 63, no. 44, p. 10840.
Tsukanov, A.V., Dubonosov, A.D., Bren’, V.A., and Minkin, V.I., Chem. Heterocycl. Comp., 2008, no. 8, pp. 899–923.
Ushakov, E.N., Alfimov, M.V., and Gromov, S.P., Russ. Chem. Rev., 2008, vol. 77, no. 1, p. 39.
Iglesias-Sanchez, J.C., Wang, W., Ferdani, R., Prados, P., de Mendoza, J., and Gokel, G.W., New J. Chem., 2008, vol. 32, no. 5, p. 878.
Yanaka, Y., Kobuke, Y., and Sokabe, M., Angew. Chem. Int. Ed., 1995, vol. 34, no. 6, p. 693.
Cacciapaglia, R., Casnati, A., DiStefano, S., Mandolini, L., Pulemili, D., Reinhoudt, D.N., Sartori, A., and Ungaro, R., Chem. Eur. J., 2004, vol. 10, no. 18, p. 4436.
Beer, P.D., Drew, M.G.B., Knubley, R.J., and Ogden, M.I., J. Chem. Soc. Dalton Trans., 1995, p. 3117.
Alekseeva, E.A., Luk’yanenko, A.P., Basok, S.S., Mazepa, A.V., and Gren’, A.I., Russ. J. Org. Chem., 2010, vol. 46, no. 9, p. 1403–1408.
Vetrogon, V.I., Lukyanenko, N.G., Schwing-Weill, M.-J., and Arnaud-Neu, F., Talanta, 1994, vol. 41, p. 2105.
Bentouhami, E., Bouet, G.M., Schwing, M.-J., and Khan, M.A., J. Solution Chem., 2006, vol. 35, p. 889.
Kinard, W.F., Grant, P.M., and Baisden, P.A., Polyhedron, 1989, no. 8, p. 2385.
Roymon, J., Balaji, R., Amitabha, A., Anupam, K., and Chebrolu, P.R., J. Org. Chem., 2008, vol. 73, no. 15, p. 5745.
Casnati, A., Barboso, S., Rouquette, H., SchwingWeill, M.-J., Arnaud-Neu, F., Dozol, J.-F., and Ungaro, R., J. Am. Chem. Soc., 2001, vol. 123, no. 49, p. 12182.
Kim, H.J., Kim, S.K., Lee, J.Y., and Kim, J.S., J. Org. Chem., 2006, vol. 71, no. 17, p. 6611.
Arnaud-Neu, F., Barrett, G., Fanni, S., Marrs, D., and McGregor, W., J. Chem. Soc., Perkin Trans. 2, 1995, no. 3, p. 453.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.A. Alekseeva, S.S. Basok, A.V. Mazepa, A.P. Luk’yanenko, O.V. Snurnikova, A.I. Gren’, 2013, published in Zhurnal Obshchei Khimii, 2013, Vol. 83, No. 9, pp. 1519–1525.
Rights and permissions
About this article
Cite this article
Alekseeva, E.A., Basok, S.S., Mazepa, A.V. et al. p-tert-butylcalix[4]arenes containing azacrown ether substituents at the lower rim as potential polytopic receptors. Russ J Gen Chem 83, 1738–1743 (2013). https://doi.org/10.1134/S1070363213090181
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363213090181