Abstract
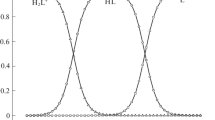
Direct calorimetric method was used to determine the heat of reaction of L-valyl-L-valine stepwise dissociation. The standard thermodynamic characteristics of the studied equilibria were established. The results are compared with the corresponding data for related compounds.
Similar content being viewed by others
References
Rodante, F. and Fantauzzi, F., Termochim. Acta, 1989, vol. 154, p. 279.
Vasil’ev, V.P., Termodinamicheskie svoistva rastvorov elektrolitov (Thermodynamic Characteristics of Electrolyte Solutions), Moscow: Vysshaya Shkola, 1982.
Petrov, N.V., Nabokov, V.S., Zhadanov, B.V., Savich, I.A., and Spitsyn, V.I., Zh. Fiz. Khim., 1976, vol. 50, no. 9, p. 2208.
Gridchin, S.N. and Gorboletova, G.G., Zh. Obshch. Khim., 2008, vol. 78, no. 3, p. 444.
Gorboletova, G.G. and Kochergina, L.A., J. Term. Anal. Cal., 2007, vol. 87, no. 2, p. 561.
Gridchin, S.N., Gorboletova, G.G., and Pyreu, D.F., Zh. Fiz. Khim., 2007, vol. 81, no. 12, p. 2165.
Gorboletova, G.G., Gridchin, S.N., and Sazonova, E.S., Zh. Fiz. Khim., 2005, vol. 79, no. 8, p. 1390.
Gridchin, S.N., Gorboletova, G.G., and Pyreu, D.F., J. Term. Anal. Cal., 2007, vol. 90. No. 2, p. 607.
Vasil’ev, V.P., Kochergina, L.A., and Krutova, O.N., Izv. Vuzov, Ser. Khim. i Khim. Tekhnol., 2003, vol. 46, no. 6, p. 69.
Gridchin, S.N. and Pyreu, D.F., Zh. Obshch. Khim., 2007, vol. 77, no. 6, p. 948.
Gridchin, S.N., Zh. Obshch. Khim., 2010, vol. 80, no. 3, p. 369.
Varfolomeev, S.D., Khimicheskaya enzimologiya (Chemical Enzimology), Moscow: Akademiya, 2005.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.N. Gridchin, 2013, published in Zhurnal Obshchei Khimii, 2013, Vol. 83, No. 7, pp. 1061–1064.
Rights and permissions
About this article
Cite this article
Gridchin, S.N. Thermodynamic characteristics of protolytic equilibria of L-valyl-L-valine in aqueous solution. Russ J Gen Chem 83, 1315–1317 (2013). https://doi.org/10.1134/S1070363213070025
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363213070025