Abstract
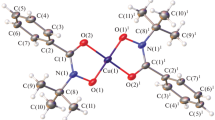
Structures of three mixed solvates of copper halides: CuBr2·DX·2H2O, CuBr2·2DX·2H2O, and CuCl2·2DX·2H2O (DX is 1,4-dioxane) has been determined by the method of XRD on single crystals. The entry of both solvents into the first coordination sphere of the copper ion was proved. In spite of analogous composition, CuHlg2·2DX·2H2O solvates have different structures: the chain (Hlg = Br) and the island (Hlg = Cl) structures. The difference is caused by weakening donor power of acido ligands on passing from Br to Cl.
Similar content being viewed by others
References
Bersuker, I.B., The Jahn-Teller Effect, Cambridge: Cambridge University Press, 2006, p. 437.
Persson, I., Persson, P., Sandstrom, M., and Ullstrom, A.-S., J. Chem. Soc. Dalton Trans., 2002, p. 1256.
Gutmann, V., Coordination Chemistry in Non-Aqueous Solutions, Wien: Springer Verlag, 1968.
X-RED Version 1.09, X-SHAPE Version 1.02, STOE and Cie GmbH, Darmstadt, 1997.
Altomare, A., Cascarano, G., Giacovazzo, C., and Gualardi, A., J. Appl. Cryst., 1993, vol. 26, p. 343.
Sheldrick, G.M., SHELX 97, University of Gottingen, Germany, 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.L. Starova, K.K. Spektor, Yu.M. Skripkin, 2011, published in Zhurnal Obshchei Khimii, 2011, Vol. 81, No. 9, pp. 1428–1431.
Rights and permissions
About this article
Cite this article
Starova, G.L., Spektor, K.K. & Skripkin, Y.M. Structure of copper dihalide-1,4-dioxane-water mixed solvates. Russ J Gen Chem 81, 1768–1771 (2011). https://doi.org/10.1134/S1070363211090052
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363211090052