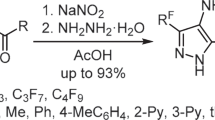
Abstract
A convenient oxidation method of 1-(2-chloroethyl)-4-formylpyrazoles followed by dehydrochlorination was developed for preparation of 1-vinyl-4-pyrazolecarboxylic acids. Dehydrochlorination rate was established to decrease with the growing number of electron-donor substituents.
Similar content being viewed by others
References
Gibson, N.A. and Hosking, J.N., Aust. J. Chem., 1965, vol. 18, no. 1, p. 123.
Yanovskaya, L.A. and Yufit, S.S., Organicheskii sintez v dvukhfaznykh sistemakh (Organic Synthesis in Two-Phase Systems), Moscow: Khimiya, 1982.
Herriot, A.W. and Picker, D., Tetrahedron Lett., 1974, no. 16, p. 1511.
Jam, D.J. and Simmons, H.E., J. Am. Chem. Soc., 1972, vol. 94, no. 11, p. 4024.
Manger, F.M., Rhee, J.U., and Rhee, H.K., J. Org. Chem., 1975, vol. 40, no. 25, p. 3803.
Attaryan, O.S., Gavalyan, V.B., Elizasyan, G.A., Asratyan, G.V., and Darbinyan, E.G., Arm. Khim. Zh., 1988, vol. 41, no. 8, p. 496.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.O. Baltayan, A.A. Saakyan, O.S. Attaryan, G.V. Asratyan, 2010, published in Zhurnal Obshchei Khimii, 2010, Vol. 80, No. 5, pp. 834–836.
Rights and permissions
About this article
Cite this article
Baltayan, A.O., Saakyan, A.A., Attaryan, O.S. et al. Synthesis of 1-vinyl-4-pyrazolecarboxylic acids. Russ J Gen Chem 80, 1004–1006 (2010). https://doi.org/10.1134/S1070363210050257
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363210050257