Abstract
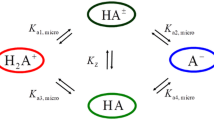
A procedure for estimation of dissociation constants of protonated tribasic species is developed distinguished by a new approach to evaluation of concentrations of all moieties in the equilibrium and accounting for it in the calculation of the solution ionic strength.
Similar content being viewed by others
References
Tanganov, B.B. and Alekseeva, I.A., Zh. Obshch. Khim., 2006, vol. 76, no. 11, p. 1800.
Tanganov, B.B. and Alekseeva, I.A., Zh. Obshch. Khim., 2005, vol. 75, no. 11, p. 1775.
Speakman, J.C., J. Chem. Soc., 1940, p. 855.
Al’bert, A. and Serzhent, E., Konstanty ionizatsii kislot i osnovanii (Ionization Constants of Acids and Bases), Moscow: Khimiya, 1964.
Beits, R., Opredelenie pH. Teoriya i praktika (Measuring of pH. Theory and Praxis), Leningrad: Khimiya, 1972.
Anorganikum, Moscow: Mir, 1984, vol. 2, p.120.
Tanganov, B.B., Khimicheskie metody analiza: Uchebnoe posobie (Kinetical Methods of Analysis. Educational Testbook), Ulan-Ude: 2005.
Davies, C.W., J. Chem. Soc., 1938, p. 2093.
Waisberger, A., Proskauer, E., Riddik, J., and Tups, E., Organicheskie rastvoriteli (Organic Solvents), Moscow: Izdatinlit, 1958.
Gordon, A.J. and Ford, R.A., The Chemist’s Companion. A Handbook of Practical Data, Techniques and References, New York: Wiley, 1972.
Tanganov, B.B., Khim. Khim. Technol., Dep. ONIITEKhim, 1984, no. 976, D84.
Aleksandrov, V.V., Lebed’, V.I., Shikhova, T.M., and Zaslavskii B.G., Elektrokhimiya, 1968, vol. 4, no. 6, p. 711.
Kreshkov, A.P., Analiticheskaya khimiya nevodnykh rastvorov (Analytical Chemistry of Non-Aqueous Solutions), Moscow: Khimiya, 1982.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © B.B. Tanganov, 2009, published in Zhurnal Obshchei Khimii, 2009, Vol. 79, No. 7, pp. 1078–1080.
For communication XXI, see [1].
Rights and permissions
About this article
Cite this article
Tanganov, B.B. Acid-base equilibriums in solutions of polybases (model and experiment): II. Thermodynamic dissociation constants of protonated trifunctional bases. Russ J Gen Chem 79, 1434–1436 (2009). https://doi.org/10.1134/S1070363209070056
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363209070056