Abstract
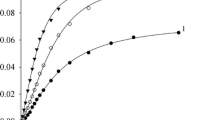
The influence of temperature on the rate of dehydrochlorination of 3-chloro-3-methylbut-1-ene in 17 aprotic and 13 protic solvents, ν = k[C5H9Cl], was studied by the verdazyl method. In aprotic solvents, the electrophilicity, ionizing power, and cohesion of solvents decrease ΔG ≠ by increasing ΔS ≠. The nucleophilicity and polarizability increase both ΔH ≠ and ΔS ≠ to equal extent and therefore do not affect ΔG ≠. In protic solvents, the solvent nucleophilicity increases ΔH ≠ to a greater extent than ΔS ≠, and the overall effect of the nucleophilic solvation is small and negative.
Similar content being viewed by others
References
Ponomarev, N.E., Zaliznyi, V.V., and Dvorko, G.F., Zh. Obshch. Khim., 2005, vol. 75, no. 10, p. 1593.
Ponomarev, N.E., Zaliznyi, V.V., and Dvorko, G.F., Zh. Obshch. Khim., 2005, vol. 75, no. 9, p. 1503.
Sneen, R.A., Carter, J.V., and Kay, P.S., J. Am. Chem. Soc., 1966, vol. 88, no. 11, p. 2594.
Dvorko, G.F., Ponomarev, N.E., and Ponomareva, E.A., Zh. Obshch. Khim., 1999, vol. 69, no. 11, p. 1835.
Dvorko, G.F., Ponomareva, E.A., and Ponomarev, M.E., J. Phys. Org. Chem., 2004, vol. 17, no. 4, p. 825.
Reichardt, Ch., Solvents and Solvent Effects in Organic Chemistry, Weinheim: VCH, 1988.
Dvorko, G.F., Pervishko, T.L., Golovko, N.I., Vasil’kevich, A.I., and Ponomareva, E.A., Zh. Org. Khim., 1993, vol. 29, no. 9, p. 1805.
Ponomarev, N.E., Stambirskii, M.V., and Dvorko, G.F., Zh. Org. Khim., 2004, vol. 40, no. 4, p. 520.
Vasil’kevich, A.I., Ponomareva, E.A., and Dvorko, G.F., Zh. Org. Khim., 1990, vol. 26, no. 11, p. 2267.
Dvorko, G.F., Zaliznyi, V.V., and Ponomarev, N.E., Zh. Obshch. Khim., 2002, vol. 72, no. 10, p. 1644.
Isaacs, N.S., Physical Organic Chemistry, New York: Wiley, 1992.
Buncel, E., Millington, J.P., and Wiltshire, J.F., Can. J. Chem., 1977, vol. 55, no. 11, p. 1401.
Quameneur, F., Baiou, B., and Kerfanto, M., C. R. Acad. Sci. Paris, 1974, vol. 278, no. 1, p. 299.
Abraham, M.H., Grellier, P.L., Nasehzadeh, A., and Walker, A.C., J. Chem. Soc., Perkin Trans. 2, 1988, no. 6, p. 1717.
Dvorko, G.F., Zaliznyi, V.V., and Ponomarev, N.E., Zh. Obshch. Khim., 2002, vol. 72, no. 9, p. 1501.
Dvorko, G.F., Zaliznyi, V.V., and Ponomarev, N.E., Zh. Obshch. Khim., 2004, vol. 74, no. 9, p. 1470.
Dvorko, G.F. and Ponomareva, E.A., Usp. Khim., 1991, vol. 60, no. 10, p. 2089.
Amelichev, V.A., Trunov, Yu.P., and Saidov, G.V., Vestn. Leningr. Gos. Univ., Ser. 2: Khim., 1974, no. 10, p. 83.
Dvorko, G.F. and Evtushenko, N.Yu., Zh. Obshch. Khim., 1991, vol. 61, no. 9, p. 205.
Koppel, I.A. and Palm, V.A., Advances in Linear Free Energy Relationship, Chapman, N.B. and Schoter, J., Eds., London: Plenum, 1972, p. 208.
Makitra, R.G. and Pirig, Ya.N., Zh. Obshch. Khim., 1986, vol. 56, no. 3, p. 657.
Kamlet, M.J. and Taft, R.W., J. Am. Chem. Soc., 1976, vol. 98, no. 2, p. 377.
Marcus, Y., Chem. Soc. Rev., 1993, vol. 22, no. 3, p. 406.
Palm, V.A., Osnovy kolichestvennoi teorii organicheskikh reaktsii (Principles of the Quantitative Theory of Organic Reactions), Leningrad: Khimiya, 1977.
Abboud, J.-L. and Notario, R., Pure Appl. Chem., 1999, vol. 71, no. 4, p. 645.
Kafarov, V.V., Metody kibernetiki v khimii i khimicheskoi tekhnologii (Cybernetic Methods in Chemistry and Chemical Technology), Moscow: Khimiya, 1971.
Gajewski, J.J., J. Am. Chem. Soc., 2001, vol. 123, no. 44, p. 10 877.
Dvorko, G.F., Koshchii, I.V., Prokopets, A.M., and Ponomareva, E.A., Zh. Obshch. Khim., 2002, vol. 72, no. 12, p. 1982.
McLennan, D.J. and Martin, P.L., J. Chem. Soc., Perkin Trans. 2, 1982, no. 6, p. 1091.
Okuno, Y., J. Phys. Chem., 1999, vol. 103, no. 11, p. 190.
Yamabe, S. and Tsuchida, N., J. Comput. Chem., 2004, vol. 25, no. 4, p. 598.
Okuno, Y., J. Am. Chem. Soc., 2000, vol. 122, no. 12, p. 2925.
Hollenstein, S. and Laube, T., J. Am. Chem. Soc., 1993, vol. 115, no. 22, p. 7240.
Jagannadham, V., Proc. Indian Acad. Sci., 2003, vol. 115, no. 1, p. 41.
Dvorko, G.F., Vasil’kevich, A.I., and Ponomarev, N.E., Zh. Org. Khim., 1997, vol. 33, no. 2, p. 245.
Dvorko, G.F., Cherevach, T.V., Kulik, N.I., and Ponomarev, N.E., Zh. Obshch. Khim., 1994, vol. 64, no. 6, p. 979.
Dvorko, G.F., Koshchii, I.V., Prokopets, A.M., and Ponomareva, E.A., Zh. Obshch. Khim., 2002, vol. 72, no. 12, p. 1989.
Koshchii, I.V., Cand. Sci. (Chem.) Dissertation, Kiev, 2002.
Ingold, C.K., Structure and Mechanism in Organic Chemistry, Ithaca: Cornell Univ. Press, 1969.
Okamoto, K., Nioto, I., Dohi, M., and Shingu, H., Bull. Chem. Soc. Jpn., 1971, vol. 44, no. 2, p. 320.
Kinoshita, T., Ueno, T., Ikai, K., Fujiwara, M., and Okamoto, K., Bull. Chem. Soc. Jpn., 1988, vol. 61, no. 12, p. 3273.
Entelis, S.G. and Tiger, R.P., Kinetika reaktsii v zhidkoi faze (Kinetics of Liquid-Phase Reactions), Moscow: Khimiya, 1973.
Gorodynskii, V.A. and Morachevskii, A.A., Dokl. Akad. Nauk SSSR, 1975, vol. 224, no. 4, p. 855.
Kim, H.J. and Hynes, J.T., J. Am. Chem. Soc., 1992, vol. 114, no. 26, p. 10 508.
Jencks, W.P., Chem. Soc. Rev., 1980, vol. 10, no. 2, p. 345.
Claesson, S., Lundgren, B., and Szwarc, M., Trans. Faraday Soc., 1970, vol. 66, no. 9, p. 3053.
Ponomareva, E.A., Pervischko, T.L., and Dvorko, G.F., Org. React. (Tartu), 1981, vol. 18, no. 2, p. 312.
Farcasin, D., Jähme, J., and Rüchardt, Ch., J. Am. Chem. Soc., 1985, vol. 107, no. 20, p. 5717.
Zillian, U., Chem. Ztg., 1984, vol. 108, no. 2, p. 381.
Katriski, A.P. and Brycki, B., J. Am. Chem. Soc., 1986, vol. 108, no. 23, p. 7295.
Winstein, S., Appel, B., Baker, K., and Diaz, L., J. Chem. Soc., Spec. Publ., 1965, no. 19, p. 109.
Dvorko, G.F. and Ponomareva, E.A., Zh. Org. Khim., 1998, vol. 34, no. 4, p. 487.
Dvorko, G.F. and Ponomarev, N.E., Zh. Obshch. Khim., 1997, vol. 67, no. 6, p. 908.
Kelly, X.H., Tanaka, S.E., and Bennet, A.J., J. Am. Chem. Soc., 1998, vol. 120, no. 7, p. 1405.
Mayr, H. and Minegishi, Sh., Angew. Chem., Int. Ed., 2002, vol. 41, no. 23, p. 4493.
Richard, J.P., Toteva, M., and Amyes, T.L., Org. Lett., 2001, vol. 3, no. 14, p. 2225.
Liu, K-I., J. Chin. Chem. Soc., 1995, vol. 42, no. 4, p. 607.
Takeuchi, K., Pure Appl. Chem., 1998, vol. 70, no. 10, p. 2023.
Bentley, T.W. and Llewellyn, G., Prog. Phys. Org. Chem., 1990, vol. 17, p. 121.
Kevill, D.N. and D’souza, M.J., J. Phys. Org. Chem., 1992, vol. 5, no. 3, p. 287.
Takeuchi, K., Takasuka, M., Shiba, E., Kinoshita, T., Okazaki, T., Abboud, J.-L., Notario, R., and Castano, O., J. Am. Chem. Soc., 2000, vol. 122, no. 30, p. 7351.
Dvorko, G.F., Koshchii, I.V., and Ponomareva, E.A., Zh. Obshch. Khim., 2003, vol. 73, no. 4, p. 603.
Dvorko, G.F., Koshchii, I.V., and Ponomareva, E.A., Zh. Obshch. Khim., 2003, vol. 73, no. 3, p. 404.
Dvorko, G.F. and Ponomar’ova, E.O., Ukr. Khim. Zh., 1993, vol. 59, no. 11, p. 1190.
Abboud, J.-L., Herreros, M., Notario, R., Lomas, J.S., Madera, J., Mullea, P., and Rossier, J.-C., J. Org. Chem., 1999, vol. 64, no. 16, p. 6401.
Abboud, J-L.M., Castano, O., Della, E.W., Herreros, M., Muller, P., Notario, R., and Rossier, J-C., J. Am. Chem. Soc., 1997, vol. 119, no. 9, p. 2262.
Takeuchi, K., Ohga, Y., Ushino, T., and Takasuka, M., J. Phys. Org. Chem., 1997, vol. 10, no. 4, p. 717.
Abboud, J.-L.M., Alkorta, I., Davalos, J.Z., Muller, P., Quintanilla, E., and Rossier, J.-C., J. Org. Chem., 2003, vol. 68, no. 10, p. 3786.
Raber, D.J., Harris, J.M., and Schleyer, P.v.R., Ions and Ion Pairs in Organic Reactions, Szwarz, M., Ed., New York: Wiley, 1974, vol. 2, p. 247.
Dvorko, G.F., Evtushenko, N.Yu., and Zhovtyak, V.N., Zh. Obshch. Khim., 1987, vol. 57, no. 5, p. 1157.
Dvorko, G.F. and Ponomareva, E.A., Org. React. (Tartu), 1985, vol. 22, no. 4, p. 451.
Sneen, R.A. and Kay, P.S., J. Am. Chem. Soc., 1972, vol. 94, no. 20, p. 6983.
Kuhn, R. and Trischman, H., Monatsh. Chem., 1964, vol. 95, no. 2, p. 457.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.E. Ponomarev, V.V. Zaliznyi, G.F. Dvorko, 2007, published in Zhurnal Obshchei Khimii, 2007, vol. 77, No. 7, pp. 1120–1130.
For communication XLII, see [1].
Rights and permissions
About this article
Cite this article
Ponomarev, N.E., Zaliznyi, V.V. & Dvorko, G.F. Kinetics and mechanism of monomolecular heterolysis of commercial organohalogen compounds: XLIII. Solvent effect on activation parameters of dehydrochlorination of 3-chloro-3-methylbut-1-ene. Correlation analysis of solvation effects. Russ J Gen Chem 77, 1204–1214 (2007). https://doi.org/10.1134/S1070363207070110
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070363207070110