Abstract
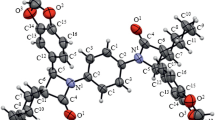
Reformatsky reagent generated from methyl 1-bromocyclohexane-1-carboxylate reacted with N-aryl-2-oxochromene-3-carboxamides and N-aryl-6-bromo-2-oxochromene-3-carboxamides to give, depending on the conditions, the corresponding N-aryl-(6-bromo)-4-(1-methoxycarbonylcyclohexyl)-2-oxochroman-3-carboxamides or 3-aryl-(9-bromo)-1,1-pentamethylene-2,3,4,4a,5,10b-hexahydro-1H-chromeno[3,4-c]pyridine-2,4,5-triones. The products were isolated as a single diastereoisomer.
Similar content being viewed by others
References
Shchepin, V.V., Fotin, D.V., Vakhrin, M.I., and Shurov, S.N., Russ. J. Gen. Chem., 2004, vol. 74, no. 9, p. 1406.
Stewart, J.J.P., J. Comput. Chem., 1989, vol. 10, no. 2, p. 209.
Karplus, M., J. Chem. Phys., 1959, vol. 30, no. 1, p. 11.
Bothner-By, A.A., Adv. Magn. Reson., 1965, vol. 1, no. 2, p. 195.
Stewart, J.J.P., Mopac 7.0, Frank J. Seiler Res. Lab. US Air Force Academy, QCPM 175.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.V. Shchepin, N.F. Kirillov, M.I. Vakhrin, O.B. Bayanova, S.N. Shurov, P.S. Silaichev, 2006, published in Zhurnal Obshchei Khimii, 2006, Vol. 76, No. 7, pp. 1194–1197.
Rights and permissions
About this article
Cite this article
Shchepin, V.V., Kirillov, N.F., Vakhrin, M.I. et al. Reformatsky reaction of methyl 1-bromocyclohexane-1-carboxylate with N-aryl-2-oxochromene-3-carboxamides. Russ J Gen Chem 76, 1146–1149 (2006). https://doi.org/10.1134/S1070363206070255
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070363206070255