Abstract
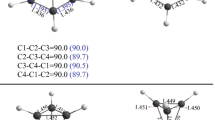
The RHF/6-311G(d) and MP2/6-311G(d) calculations with full geometry optimization were performed for CH3COX molecules (X = F, Cl, Br, CH3). Variations in the populations of the p y orbitals of their halogen and carbon atoms (orbitals whose symmetry axes are perpendicular to the molecular plane) from X = F to X = Cl, Br, and CH3 are not associated with variations in the extent of the p,π conjugation between the lone electron pair of the halogen atom and the π-electron system of the carbonyl group. The bonding molecular orbitals formed by these atomic p y orbitals are not determined by this interaction. The RHF/6-311G(d) and MP2/6-311G(d) calculations give similar results.
Similar content being viewed by others
References
Feshin, V.P., Feshina, E.V., and Zhizhina, L.I., Zh. Obshch. Khim., 2006, vol. 76, no. 5, p. 770.
Lucken, E.A.C., Nuclear Quadrupole Coupling Constants, New York: Academic, 1969.
Hart, R.M. and Whitehead, M.A., Can. J. Chem., 1971, vol. 49, no. 15, p. 2508.
Voronkov, M.G. and Feshin, V.P., Org. Magn. Reson., 1977, vol. 9, no. 12, p. 665.
Feshin, V.P., Voronkov, M.G., Dolgushin, G.V., Nikitin, P.A., Lazarev, I.M., Sapozhnikov, Yu.E., and Yasman, Ya.B., Dokl. Akad. Nauk SSSR, 1983, vol. 268, no. 5, p. 1163.
Feshin, V.P. and Konshin, M.Yu., J. Mol. Struct., 1995, vol. 345, no. 1, p. 83.
Feshin, V.P., Elektronnye effekty v organicheskikh i elementoorganicheskikh molekulakh (Electronic Effects in Organic and Organoelement Molecules), Yekaterinburg: Urasl’sk. Otd. Ross. Akad. Nauk, 1997.
Feshin, V.P. and Feshina, E.V., Z. Naturforsch., A, 2000, vol. 55, no. 2, p. 555.
Shlyapnikov, D.B. and Feshin, V.P., Z. Naturforsch., A, 2002, vol. 57, no. 4, p. 974.
Feshin, V.P. and Shlyapnikov, D.B., Z. Naturforsch., A, 2003, vol. 58, no. 2, p. 475.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Gill, P.M.W., Johnson, B.G., Robb, M.A., Cheeseman, J.R., Keith, T., Petersson, G.A., Montgomery, J.A., Raghavachari, K., Al-Laham, M.A., Zakrzewski, V.G., Ortiz, J.V., Foresman, J.B., Cioslowski, J., Stefanov, B.B., Nanayakkara, A., Challacombe, M., Peng, C.Y., Ayala, P.Y., Chen, W., Wong, M.W., Andres, J.L., Replogle, E.S., Gomperts, R., Martin, R.L., Fox, D.J., Binkley, J.S., Defrees, D.J., Baker, J., Stewart, J.P., Head-Gordon, M., Gonzalez, C., and Pople, J.A., GAUS-SIAN-94. Rev. E.3, Pittsburgh: Gaussian, 1995.
Pierce, L. and Krisher, L.C., J. Chem. Phys., 1959, vol. 31, no. 4, p. 875.
Tsuchiya, S., J. Mol. Struct., 1974, vol. 22, no. 1, p. 77.
Tsuchiya, S. and Kimura, M., Bull. Chem. Soc. Jpn., 1972, vol. 45, no. 3, p. 736.
Tsuchiya, S. and Iijima, T., J. Mol. Struct., 1972, vol. 13, no. 2, p. 327.
Feshin, V.P. and Kon’shin, M.Yu., Zh. Obshch. Khim., 1996, vol. 66, no. 6, p. 951.
Foresman, J.B. and Frisch, A.E., Exploring Chemistry with Electronic Structure Methods, Pittsburgh: Gaussian, 1996.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.P. Feshin, E.V. Feshina, L.I. Zhizhina, 2006, published in Zhurnal Obshchei Khimii, 2006, Vol. 76, No. 5, pp. 776–779.
For communication I, see [1].
Rights and permissions
About this article
Cite this article
Feshin, V.P., Feshina, E.V. & Zhizhina, L.I. An ab initio evaluation of the role of p,π interaction: II. Molecules of the CH3COX series. Russ J Gen Chem 76, 739–742 (2006). https://doi.org/10.1134/S1070363206050148
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070363206050148