Abstract
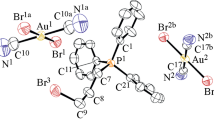
The ionic complexes [Ph3PR][Au(CN)2Cl2] (R = CH2CH=CHCH3 (I), CH2CN (II)) were prepared by the reactions of organyltriphenylphosphonium chlorides with potassium dichlorodicyanoaurate in water followed by recrystallization from acetonitrile. Apart from the major product II, the crystals of the molecular complex Ph3PC(H)(CN)Au(CN)2Cl (III) were isolated. The products were characterized by X-ray diffraction (CIF files CCDC nos. 1957185 (I), 2060227 (II), 2066549 (III)) and NMR and IR spectroscopy. According to X-ray diffraction data, complexes I and II consisted of organyltriphenylphosphonium cations with a slightly distorted tetrahedral geometry of phosphorus atoms and the centrosymmetric square anions [Au(CN)2Cl2]−, which, in the case of complex II, form coordination pseudopolymer chains via the Au∙∙∙Cl interanion contacts (3.40 Å). In complex III, the phosphorus and gold atoms are also coordinated in the tetrahedral and square environments; the ylide carbon atom is located at the gold atom in the trans-position relative to chlorine.




Similar content being viewed by others
REFERENCES
Janiak, C., Dalton Trans., 2003, no. 14, p. 2781. https://doi.org/10.1039/B305705B
Desiraju, G.R., Angew. Chem., Int. Ed., 2007, vol. 46, no. 44, p. 8342. https://doi.org/10.1002/anie.200700534
Dunitz, J.D. and Gavezzotti, A., Chem. Soc. Rev., 2009, vol. 38, no. 9, p. 2622. https://doi.org/10.1039/B822963P
Sculfort, S. and Braunstein, P., Chem. Soc. Rev., 2011, vol. 40, no. 5, p. 2741. https://doi.org/10.1039/C0CS00102C
Alkorta, I., Elguero, J., and Frontera, A., Crystals, 2020, vol. 10, no. 3, p. 180. https://doi.org/10.3390/cryst10030180
Rodina, T.A., Loseva, O.V., and Ivanov, A.V., J. Struct. Chem., 2021, vol. 62, p. 123. https://doi.org/10.1134/S0022476621010157
Batten, S.R. and Champness, N.R., Phil. Trans. R. Soc. A, 2017, vol. 375, no. 2084, p. 20160032.https://doi.org/10.1098/rsta.2016.0032
Furukawa, H., Cordova, K.E., O’Keeffe, M., and Yaghi, O.M., Science, 2013, vol. 341, no. 6149, ID 1230444. https://doi.org/10.1126/science.1230444
Liu, J., Chen, L., Cui, H., et al., Chem. Soc. Rev., 2014, vol. 43, no. 16, p. 6011. https://doi.org/10.1039/C4CS00094C
Liu, J.-Q., Luo, Z.-D., Pan, Y., et al., Coord. Chem. Rev., 2020, vol. 406, p. 213145. https://doi.org/10.1016/j.ccr.2019.213145
Baranov, A.Yu., Rakhmanova, M.I., Samsonenko, D.G., et al., Inorg. Chim Acta, 2019, vol. 494, p. 78. https://doi.org/10.1016/j.ica.2019.05.015
Petrovskii, S.K., Paderina, A.V., Sizova, A.A., et al., Dalton Trans., 2020, vol. 49, no. 38, p. 13430. https://doi.org/10.1039/D0DT02583F
Kumar, K., Stefañczyk, O., Chorazy, S., et al., Inorg. Chem., 2019, vol. 58, no. 9, p. 5677. https://doi.org/10.1021/acs.inorgchem.8b03634
Nicholas, A.D., Bullard, R.M., Pike, R.D., and Patterson, H., Eur. J. Inorg. Chem., 2019, vol. 2019, no. 7, p. 956. https://doi.org/10.1002/ejic.201801407
Belyaev, A., Eskelinen, T., Dau, T., et al., Chem.-Eur. J., 2017, vol. 24, no. 6, p. 1404. https://doi.org/10.1002/chem.201704642
Ovens, J.S., Christensen, P.R., and Leznoff, D.B., Chem.-Eur. J., 2016, vol. 22, no. 24, p. 8234. https://doi.org/10.1002/chem.201505075
Ovens, J.S., Geisheimer, A.R., Bokov, A.A., et al., Inorg. Chem., 2010, vol. 49, no. 20, p. 9609. https://doi.org/10.1021/ic101357y
Katz, M.J. and Leznoff, D.B., J. Am. Chem. Soc., 2009, vol. 131, no. 51, p. 18435. https://doi.org/10.1021/ja907519c
Thompson, J.R., Goodman-Rendall, K.A.S., and Leznoff, D.B., Polyhedron, 2016, vol. 108, p. 93. https://doi.org/10.1016/j.poly.2015.12.026
Lefebvre, J., Korčok, J.L., Katz, M.J., and Leznoff, D.B., Sensors, 2012, vol. 12, no. 3, p. 3669. https://doi.org/10.3390/s120303669
Varju, B.R., Ovens, J.S., and Leznoff, D.B., Chem. Commun., 2017, vol. 53, no. 48, p. 6500. https://doi.org/10.1039/C7CC03428H
Ovens, J.S. and Leznoff, D.B., Chem. Mater., 2015, vol. 27, no. 5, p. 1465. https://doi.org/10.1021/cm502998w
Ovens, J.S. and Leznoff, D.B., Inorg. Chem., 2017, vol. 56, no. 13, p. 7332. https://doi.org/10.1021/acs.inorgchem.6b03153
Ovens, J.S. and Leznoff, D.B., CrystEngComm, 2018, vol. 20, no. 13, p. 1769. https://doi.org/10.1039/C7CE02167D
Lefebvre, J., Chartrand, D., and Leznoff, D.B., Polyhedron, 2007, vol. 26, nos. 9−11, p. 2189. https://doi.org/10.1016/j.poly.2006.10.045
Lefebvre, J., Tyagi, P., Trudel, S., et al., Inorg. Chem., 2009, vol. 48, no. 1, p. 55. https://doi.org/10.1021/ic801094m
Geisheimer, A.R., Huang, W., Pacradouni, V., et al., Dalton Trans., 2011, vol. 40, no. 29, p. 7505. https://doi.org/10.1039/C0DT01546F
Sharutin, V.V., Sharutina, O.K., Tarasova, N.M., and Efremov, A.N., Russ. J. Inorg. Chem., 2020, vol. 65, no. 2, p. 169. https://doi.org/10.1134/S0036023620020151
Sharutin, V.V., Sharutina, O.K., Efremov, A.N., and Eltsov, O.S., Russ. J. Coord. Chem., 2020, vol. 46, no. 9, p. 631. https://doi.org/10.1134/S1070328420090031
Sharutin, V.V., Bull. South Ural State Univ., Ser. Chem., 2020, vol. 12, no. 2, p. 74. https://doi.org/10.14529/chem200208
Sharutin, V.V., Sharutina, O.K., Tarasova, N.M., et al., Russ. Chem. Bull., 2020, vol. 69, no. 10, p. 1892. https://doi.org/10.1007/s11172-020-2975-4
Efremov, A.N., Sharutin, V.V., Sharutina, O.K., et al., Izv. Vyssh. Uchebn. Zaved., Ser. Khim. Tekhnol., 2020, vol. 63, no. 3, p. 10. https://doi.org/10.6060/ivkkt.20206303.6097
Shevchenko, D.P. and Khabina, A.E., Bull. South Ural State Univ. Ser. Chem., 2021, vol. 13, no. 1, p. 58. https://doi.org/10.14529/chem210106
Johnson, A., Doctoral Thesis, Zaragoza: Zaragoza University, 2018, p. 401.
SMART and SAINT-Plus. Versions 5.0. Data Collection and Processing Software for the SMART System, Madison: Bruker AXS Inc., 1998.
SHELXTL/PC. Versions 5.10. An Integrated System for Solving, Refining and Displaying Crystal Structures from Diffraction Data, Madison: Bruker AXS Inc., 1998.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339. https://doi.org/10.1107/S0021889808042726
Vicente, J. and Chicote, M.T., Coord. Chem. Rev., 1999, vols. 193–195, p. 1143. https://doi.org/10.1016/S0010-8545(99)00083-1
Djordjevic, B., Schuster, O., and Schmidbaur, H., Z. Naturforsch., A: Phys. Sci., 2005, vol. 60, no. 2, p. 169. https://doi.org/10.1039/C3CY00240C
Ahlsten, N., Perry, G.J.P., Cambeiro, X.C., et al., Catal. Sci. Technol., 2013, vol. 3, no. 11, p. 2892. https://doi.org/10.1515/znb-2005-0207
Pretsch, E., Bühlmann, P., Affolter, C., Structure Determination of Organic Compounds. Tables of Spectral Data, Berlin: Springer, 2000.
Cordero, B., Gómez, V., Platero-Prats, A.E., et al., Dalton Trans., 2008, no. 21, p. 2832. https://doi.org/10.1039/B801115J
Johnson, A., Marzo, I., and Gimeno, M.C., Chem.-Eur. J., 2018, no. 45, p. 11693. https://doi.org/10.1002/chem.201801600
Mantina, M., Chamberlin, A.C., Valero, R., et al., J. Phys. Chem. A, 2009, vol. 113, no. 19, p. 5806. https://doi.org/10.1021/jp8111556
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Rights and permissions
About this article
Cite this article
Shevchenko, D.P., Khabina, A.E., Sharutin, V.V. et al. Synthesis and Structure of Gold Complexes [Ph3PR][Au(CN)2Cl2] (R = CH2CH=CHCH3, CH2CN) and Ph3PC(H)(CN)Au(CN)2Cl. Russ J Coord Chem 48, 26–32 (2022). https://doi.org/10.1134/S1070328422010055
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328422010055