Abstract
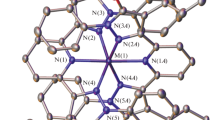
The reaction of a new tridentate ligand 2,6-bis(5-tert-butyl-1-(2,6-difluorophenyl)-1H-pyrazol-3-yl)pyridine (L) with the divalent iron salt affords the iron(II) complex [Fe(L)2](BF4)2 (I), which is isolated in the individual state and characterized by elemental analysis, NMR spectroscopy, and X-ray diffraction analysis. According to the X-ray diffraction results and data of the Evans method, which makes it possible to determine the spin state of paramagnetic compounds in a solution from the NMR spectra, the iron(II) ion in complex I exists in the high-spin state (S = 2 for Fe(II)) and undergoes no temperature-induced spin transition in a range of 120–345 K.



Similar content being viewed by others
REFERENCES
Spin-Crossover Materials: Properties and Applications, Halcrow M.A., Ed., New York: Wiley, 2013.
König, E., Progress in Inorganic Chemistry, 2007, p. 527.
Bousseksou, A., Molnár, G., Demont, P., and Menegotto, J., J. Mat. Chem., 2003, vol. 13, no. 9, p. 2069.
Matsuda, M. and Tajima, H., Chem. Lett., 2007, vol. 36, no. 6, p. 700.
Kahn, O. and Martinez, C.J., Science, 1998, vol. 279, no. 5347, p. 44.
Matsuda, M., Isozaki, H., and Tajima, H., Thin Solid Films, 2008, vol. 517, no. 4, p. 1465.
Phan, H., Hrudka, J.J., Igimbayeva, D., et al., J. Am. Chem. Soc., 2017, vol. 139, no. 18, p. 6437.
Rodríguez-Jimenéz, S., Yang, M., Stewart, I., et al., J. Am. Chem. Soc., 2017, vol. 139, no. 50, p. 18392.
Kimura, A. and Ishida, T., ACS Omega, 2018, vol. 3, no. 6, p. 6737.
Kershaw Cook, L.J., Kulmaczewski, R., Mohammed, R., et al., Angew. Chem., Int. Ed. Engl., 2016, vol. 55, no. 13, p. 4327.
Elhaïk, J., Evans, D.J., Kilner, C.A., and Halcrow, M.A., Dalton Trans., 2005, no. 9, p. 1693.
McPherson, J.N., Elton, T.E., and Colbran, S.B., Inorg. Chem., 2018, vol. 57, no. 19, p. 12312.
Santoro, A., Kershaw Cook, L.J., Kulmaczewski, R., et al., Inorg. Chem., 2015, vol. 54, no. 2, p. 682.
Nakano, K., Suemura, N., Yoneda, K., et al., Dalton Trans., 2005, no. 4, p. 740.
Kitchen, J.A., Olguín, J., Kulmaczewski, R., et al., Inorg. Chem., 2013, vol. 52, no. 19, p. 11185.
Hoselton, M., Wilson, L.J., and Drago, R.S., J. Am. Chem. Soc., 1975, vol. 97, no. 7, p. 1722.
Halcrow, M.A., Crystals, 2016, vol. 6, no. 5, p. 58.
Halcrow, M.A., Coord. Chem. Rev., 2009, vol. 253, no. 21, p. 2493.
Halcrow, M.A., Coord. Chem. Rev., 2005, vol. 249, no. 25, p. 2880.
Scudder, M.L., Craig, D.C., and Goodwin, H.A., CrystEngComm, 2005, vol. 7, no. 107, p. 642.
Clemente-León, M., Coronado, E., Giménez-López, M.C., and Romero, F.M., Inorg. Chem., 2007, vol. 46, no. 26, p. 11266.
Coronado, E., Giménez-López, M.C., Gimenez-Saiz, C., and Romero, F.M., CrystEngComm, 2009, vol. 11, no. 10, p. 2198.
Bartual-Murgui, C., Codina, C., Roubeau, O., and Aromi, G., Chem.-Eur. J., 2016, vol. 22, no. 36, p. 12767.
Jornet-Molla, V., Gimenez-Saiz, C., and Romero, F.M., Crystals, 2018, vol. 8, p. 439.
Nikovskiy, I., Polezhaev, A., Novikov, V., et al., Chem.-Eur. J., 2020. https://doi.org/10.1002/chem.202000047
van der Valk, P. and Potvin, P.G., Org. Chem., 1994, vol. 59, no. 7, p. 1766.
Evans, D.F., J. Chem. Soc. (Resumed), 1959, p. 2003.
Polezhaev, A.V., Chen, C.-H., Kinne, A.S., et al., Inorg. Chem., 2017, vol. 56, no. 16, p. 9505.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Bain, G.A. and Berry, J.F., J. Chem. Educ., 2008, vol. 85, no. 4, p. 532.
Korzekwa, J., Scheurer, A., Heinemann, F.W., and Meyer, K., Dalton Trans., 2017, vol. 46, no. 40, p. 13811.
Alvarez, S., J. Am. Chem. Soc., 2003, vol. 125, no. 22, p. 6795.
Nelyubina, Y.V., Polezhaev, A.V., Pavlov, A.A., et al., Magnetochemistry, 2018, vol. 4, no. 4, p. 46.
Alvarez, S., Chem. Rev., 2015, vol. 115, p. 13447.
Kershaw, Cook, L., Mohammed, R., Sherborne, G., et al., Coord. Chem. Rev., 2015, vols. 289–290, p. 2.
Funding
The XRD studies of the metal complexes were collected with a support from the Ministry of Science and Higher Education of the Russian Federation using the scientific equipment of the Center of Molecular Structure Investigation at the Nesmeyanov Institute of Organoelement Compounds (Russian Academy of Sciences).
This work was supported by the Russian Science Foundation, project no. 17-13-01456.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
The authors congratulate Academician I.L. Eremenko with a 70th birthday
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Nikovskii, I.A., Polezhaev, A.V., Aleshin, D.Y. et al. Synthesis and Spin State of the Iron(II) Complex with the N,N'-Disubstituted 2,6-Bis(pyrazol-3-yl)pyridine Ligand. Russ J Coord Chem 46, 402–410 (2020). https://doi.org/10.1134/S107032842006007X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107032842006007X