Abstract
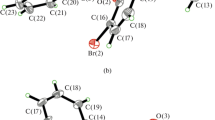
Two new oxidovanadium(V) complexes, [VOLL1] (I) and [VOLL2] (II), where L is the dianionic form of the hydrazone ligand 2-cyano-N'-(2-hydroxy-3-methoxybenzylidene)acetohydrazide (H2L), L1 and L2 are the deprotonated form of 2-hydroxybenzohydroxamate (HL1) and 3-hydroxy-2-methyl-4H-pyran-4-one (HL2), respectively, have been prepared and characterized by elemental analyses, IR, UV-Vis, 1H NMR and single crystal X-ray crystallographic determination (СIF files CCDC nos. 1880992 (I) and 1880993 (II)). The V atoms in the complexes are in octahedral coordination with the hydrazone ligand coordinates to the metal atoms through the phenolate O, imino N and enolate O atoms, and with 2-hydroxybenzohydroxamate and 3-hydroxy-2-methyl-4H-pyran-4-one through the carbonyl and hydroxyl O atoms. The effect of the compounds on the antimicrobial activity against Staphylococcus aureus, Escherichia coli, and Candida albicans was studied.


Similar content being viewed by others
REFERENCES
Burlov, A.S., Vlasenko, V.G., Koshchienko, Y.V., et al., Polyhedron, 2018, vol. 154, p. 65.
Salman, Y., Barlas, F.B., Yavuz, M., et al., Inorg. Chim. Acta, 2018, vol. 483, p. 98.
Muche, S., Harms, K., Biernasiuk, A., et al., Polyhedron, 2018, vol. 151, p. 465.
Valentova, J., Varenyi, S., Herich, P., et al., Inorg. Chim. Acta, 2018, vol. 480, p. 16.
Soliman, S.M., El-Faham, A., Elsilk, S.E., et al., Inorg. Chim. Acta, 2018, vol. 479, p. 275.
Nithya, P., Rajamanikandan, R., Simpson, J., et al., Polyhedron, 2018, vol. 145, p. 200.
Jana, K., Das, S., Maity, T., et al., J. Coord. Chem., 2018, vol. 71, no. 10, p. 1497.
Qian, H.Y., Russ. J. Coord. Chem., 2018, vol. 44, no. 1, p. 32.
Ribeiro, N., Roy, S., Butenko, N., et al., J. Inorg. Biochem., 2017, vol. 174, p. 63.
Silver, M.A., Cary, S.K., Stritzinger, J.T., et al., Inorg. Chem., 2016, vol. 55, no. 11, p. 5092.
Orlowska, E., Roller, A., Wiesinger, H., et al., RSC Adv., 2016, vol. 6, no. 46, p. 40238.
Vishwakarma, P.K., Mir, J.M., and Maurya, R.C., J. Chem. Sci., 2016, vol. 128, no. 4, p. 511.
Failes, T.W., Hall, M.D., and Hambley, T.W., Dalton Trans., 2003, no. 8, p. 1596.
Sheng, G.H., Huo, Y., Ye, Y.T., et al., Russ. J. Coord Chem., 2014, vol. 40, no. 9, p. 664. https://doi.org/10.1134/S1070328414090085
Sharma, N., Kumari, M., Kumar, V., et al., J. Coord. Chem., 2010, vol. 63, no. 11, p. 1940.
Soujanya, M., Rajitha, G., Umamaheswari, A., et al., Lett. Drug Des. Discov., 2018, vol. 15, no. 8, p. 875.
Kumar, S.S., Biju, S., and Sadasivan, V., J. Mol. Struct., 2018, vol. 1156, p. 201.
SMART and SAINT. Area Detector Control and Integration Software, Madison: Bruker Analytical X-ray Instruments Inc., 1997.
Sheldrick, G.M., SADABS, Program for Empirical Absorption Correction of Area Detector Data, Göttingen: Univ. of Göttingen, 1997.
North, A.C.T., Phillips, D.C., and Mathews, F.S., Acta Crystallogr., Sect. A, 1968, vol. 24, no. 3, p. 351.
Sheldrick, G.M., SHELXL-97, Program for the Refinement of Crystal Structures, Göttingen: Univ. of Göttingen, 1997.
Sarkar, A. and Pal, S., Polyhedron, 2006, vol. 25, no. 7, p. 1689.
Sarkar, A. and Pal, S., Inorg. Chim. Acta, 2008, vol. 361, no. 8, p. 2296.
Asgedom, G., Sreedhara, A., Kivikoski, J., et al., Dalton Trans., 1996, no. 1, p. 93.
Guo, S.H., Sun, N., Ding, Y.W., et al., Z. Anorg. Allg. Chem., 2018, vol. 644, no. 19, p. 1172.
Li, L., Lv, K.-W., Li, Y.-T., et al., Chin. J. Inorg. Chem., 2017, vol. 33, no. 5, p. 905.
Rosu, T., Negoiu, M., and Pasculescu, S., Eur. J. Med. Chem., 2010, vol. 45, no. 2, p. 774.
Searl, J.W., Smith, R.C., and Wyard, S.J., Proc. Phys. Soc. London, 1961, vol. 78, no. 505, p. 1174.
Funding
This research was supported by the Top-class foundation of Pingdingshan University (nos. PXY-BSQD-2018006 and PXY-PYJJ-2018002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, X.Q., Han, Y.J. & Xue, L.W. Synthesis, Characterization, Crystal Structures and Antimicrobial Activity of Oxidovanadium(V) Complexes with Mixed Ligands. Russ J Coord Chem 46, 58–63 (2020). https://doi.org/10.1134/S1070328420010054
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328420010054