Abstract
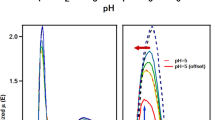
Tridentate azomethine ligands (H2L) based on 3-allylsalicylaldehyde and o-aminophenol, o-aminothiophenol, or 2-ethylamino-5-nitroaniline were synthesized and identified by IR and 1H NMR spectroscopies. The structure of complexes (Ia-Ic) was studied by magnetochemistry and EXAFS spectroscopy. Complexes Ia and Ib (X = O, S) are binuclear and exhibit antiferromagnetic exchange interaction: 2J = −36.5 and −950 cm−1, respectively. Complex Ic (X = NC2H5) is mononuclear (μeff = 1.75 μB at 275 K and does not change as temperature decreases).
Similar content being viewed by others
References
Holm, R.H. and O’Connor, M.J., Prog. Inorg. Chem., 1966, vol. 7, p. 83.
Vigato, P.A., Peruzzo, V., and Tamburini, S., Coord. Chem. Rev., 2012, vol. 256, p. 953.
Vigato, P.A., Peruzzo, V., and Tamburini, S., Coord. Chem. Rev., 2004, vol. 248, p. 1717.
Vigato, P.A., Peruzzo, V., Tamburini, S., et al., Coord. Chem. Rev., 2007, vol. 251, p. 1331.
Vigato, P.A., Peruzzo, V., and Tamburini, S., Coord. Chem. Rev., 2008, vol. 252, p. 1871.
Kogan, V.A., Zelentsov, V.V., Osipov, O.A., et al., Usp. Khim., 1979, vol. 48, no. 7, p. 1208.
Kogan, V.A., Lukov, V.V., and Shcherbakov, I.N., Russ. J. Coord. Chem., 2010, vol. 36, no. 6, p. 401.
Ovcharenko, V.I. and Sagdeev, R.Z., Usp. Khim., 1999, vol. 68, no. 5, p. 381.
Kalinnikov, V.T., Rakitin, Yu.V., and Novotortsev, V.M., Usp. Khim., 2003, vol. 72, no. 12, p. 1123.
Magnetic Molecular Magnetism, Gatteshi, D., Kahn, O., Miller, J.S., and Palacio, F., Eds., Dordrecht: Kluwer Acad., 1985.
Kahn, O., Molecular Magnetism, New York: Verlag-Chem., 1993.
Magnetism: Molecules to Material, Miller, J.S. and Drillon, V., Eds., Berlin: Verlag-Chem., 2001–2004, vols. 1–5.
Chaudhury, P., Coord. Chem. Rev., 2003, vol. 243, no. 1, p. 143.
Kahn, O., Galy, J., Journaux, Y., et al., J. Am. Chem. Soc., 1982, vol. 104, no. 8, p. 2165.
Garnovskii, A.D., Burlov, A.S., Garnovskii, D.A., et al., Polyhedron, 1999, vol. 18, p. 863.
Uraev, A.I., Vasilchenko, I.S., Ikorskii, V.N., et al., Mendeleev Commun., 2005, vol. 15, no. 4, p. 133.
Burlov, A.S., Koshchienko, Yu.V., Ikorskii, V.N., et al., Russ. J. Inorg. Chem., 2006, vol. 51, no. 7, p. 1065.
Garnovskii, A.D., Ikorskii, V.N., Uraev, A.I., et al., J. Coord. Chem., 2007, vol. 60, no. 14, p. 1493.
Burlov, A.S., Koshchienko, Yu.V., Lyssenko, K.A., et al., J. Coord. Chem., 2008, vol. 61, no. 1, p. 85.
Burlov, A.S., Nikolaevskii, S.A., Bogomyakov, A.S., et al., Russ. J. Coord. Chem., 2009, vol. 35, no. 7, p. 486.
Garnovskii, A.D., Burlov, A.S., Vasil’chenko, I.S., et al., Russ. J. Coord. Chem., 2010, vol. 36, no. 2, p. 81.
Shmakova, T.O., Garnovskii, D.A., Lysenko, K.A., et al., Russ. J. Coord. Chem., 2009, vol. 35, no. 9, p. 657.
Shmakova, T.O., Garnovskii, D.A., Lysenko, K.A., et al., Izv. Ross. Akad. Nauk., Ser. Khim., 2009, no. 7, p. 1344.
Kochubei, D.I., Babanov, Yu.A., Zamaraev, K.I., et al., Rentgenospektral’nyi metod izucheniya struktury amorfnykh tel: EXAFS-spektroskopiya (X-Ray Spectral Investigation of Solids: EXAFS Spectroscopy), Novosibirsk: Nauka, 1988.
Newville, M., J. Synchrotron Rad., 2001, vol. 8, p. 322.
Zabinski, S.I., Rehr, J.J., Ankudinov, A., and Alber, R.C., Phys. Rev., B, 1995, vol. 52, p. 2995.
Allen, F.H., Acta Crystallogr., Sect. B: Struct. Sci., 2002, vol. 58, p. 380.
Bleaney, B. and Bowers, K.D., Proc. R. Soc. London A, 1952, vol. 214, p. 451.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.S. Burlov, V.G. Vlasenko, Ya.V. Zubavichus, S.I. Levchenkov, Yu.V. Koshchienko, A.S. Bogomyakov, S.A. Nikolaevskii, D.A. Garnovskii, A.I. Uraev, T.V. Lifintseva, E.V. Korshunova, 2013, published in Koordinatsionnaya Khimiya, 2013, Vol. 39, No. 4, pp. 238–243.
Rights and permissions
About this article
Cite this article
Burlov, A.S., Vlasenko, V.G., Zubavichus, Y.V. et al. Synthesis, X-ray spectral, and magnetochemical study of copper complexes based on tridentate azomethines of 3-allylsalicylaldehyde. Russ J Coord Chem 39, 347–352 (2013). https://doi.org/10.1134/S1070328413040039
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328413040039