Abstract
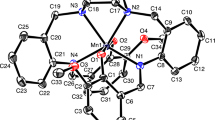
A novel binuclear Cobalt(II) complex with N-(2-propionic acid)-salicyloyl hydrazone (C10H10N2O4, H3L) was prepared and characterized. The crystal structure of [Co(C10H9N2O4)2] · 3H2O was determined by X-ray single-crystal diffractometry. The Co2+ ion is six-coordinated by the carboxyl and acyl O atoms and azomethine N atoms of two tridentate N-(2-propionicacid)-salicyloyl hydrazone ligands, which form two stable five-numbered rings sharing one side in the keto form. The coordination environment around the Co2+ ion might be described as a distorted octahedron. Abundant hydrogen bonds of the types O-H…N and O-H…O between the water molecules and ligands not only form the three-dimensional network, but also provide an extrastability for the crystal. The complex was studied for the interaction with calf thymus DNA by electronic absorption titration and emission titration. The results show that the complex is bound to calf thymus DNA mainly by intercalation.
Similar content being viewed by others
References
Wu, B.Y., Gao, L.H., Duan, Z.M., and Wang, K.Z., J. Inorg. Biochem., 2005, vol. 99, p. 1685.
Erkkila, K.E., Odom, D.T., and Barton, J.K., Chem. Rev., 1999, vol. 99, p. 2777.
Metcalfe, C. and Thomas, J.A., Chem. Soc. Rev., 2003, vol. 32, p. 215.
Haq, I., Lincoln, P., Suh, D., et al., J. Am. Chem. Soc., 1995, vol. 117, p. 4788.
Arturo, S., Giampaolo, B., Giuseppe, R., et al., J. Inorg. Biochem., 2004, vol. 98, p. 589.
Erkkila, K.E., Odom, D.T., and Barton, J.K., Chem. Rev., 1999, vol. 99, p. 2777.
Kumar, C.V., Barton, J.K., and Turro, N.J., J. Am. Chem. Soc., 1985, vol. 107, p. 5518.
Xu, H., Zheng, K.C., Chen, Y., et al., Dalton Trans., 2003, vol. 11, p. 2260.
Mahadvan, S. and Palaniandavar, M., Inorg. Chim. Acta, 1997, vol. 254, p. 291.
Xu, H., Zheng, K.C., Deng, H., et al., New J. Chem., 2003, vol. 27, no. 8, p. 1255.
Asadi, M., Safaei, E., Ranjbar, B., and Hasani, L., New J. Chem., 2004, vol. 28, no. 10, p. 1227.
Chaires, J.B., Biopolymers, 1998, vol. 44, p. 201.
Chen, F.Y., Cao, W.K., Liu, X.R., et al., Synth. React. Inorg. Met-Org. Nano-Met. Chem., 2006, vol. 36, p. 569.
Paschalidis, D.G., Tossidis, I.A., and Gdaniec, M., Polyhedron, 2000, vol. 19, p. 2629.
Yang, Z.Y., Yang, R.D., Li, F.S., et al., Polyhedron, 2000, vol. 19, p. 2599.
Deng, R.W., Wu, J.G., and Chen, Z.N., Synth. React. Met.-Org. Chem., 1992, vol. 22, p. 1295.
Singh, P.K. and Singh, B., Indian J. Chem., Sect. A: Inorg., Bio-inorg., Phys., Theor. Anal. Chem., 1998, vol. 37, p. 331.
Shen, X., Xie, Y.Y., and Jiang, H.L., Synth. React. Inorg. Met.-Org. Chem., 1995, vol. 25, p. 511.
Wang, M.X., Den, H.Q., and Shan, W.D., Chin. Chem. Lett., 1991, vol. 2, p. 587.
He, S.Y., Liu, Y., Zhao, J.S., et al., Chin. J. Chem., 2003, vol. 21, p. 139.
Satyanarayana, S., Dabrowiak, J.C., and Chaires, J.B., Biochemistry, 1992, vol. 31, p. 9319.
Wang, Y. and Yang, Z.Y., Transition Met. Chem., 2005, vol. 30, p. 902.
Li, H.B., Wang, X.P., Liu, H.P., et al., Chin. J. Inorg. Chem., 2006, vol. 22, p. 9.
He, S.Y., Liu, Y., Zhao, J.S., et al., Chin. J. Chem., 2003, vol. 21, p. 2.
Geary, W.J., Coord. Chem. Rev., 1971, vol. 7, p. 81.
Hecht, S.M. and Nat, J., J. Nat. Prod., 2000, vol. 63, no. 1, p. 158.
Bloomfield, V.A., Crothers, D.M., and Tinico, I.J., Physical Chemistry of Nucleic Acids, New York: Harper and Row, 1974, p. 187.
Wilson, W.D., Ratmeyer, L., Zhao, M., et al., Biochemistry, 1993, vol. 32, p. 4098.
Li, H., Le, X.Y., Pang, D.W., et al., J. Inorg. Biochem., 2005, vol. 99, p. 2240.
Kumar, C.V., Barton, J.K., and Turro, M.J., J. Am. Chem. Soc., 1985, vol. 107, p. 5518.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Liu, F., Zhang, WP., He, SY. et al. Synthesis, crystal structure and DNA-binding studies of the complex [Co(C10H9N2O4)2] · 3H2O. Russ J Coord Chem 35, 454–459 (2009). https://doi.org/10.1134/S1070328409060116
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328409060116