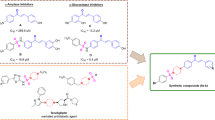
Abstract
In this research, a series of ampyrone-based Schiff base derivatives bearing the biologically active an aryl sulfonate moiety (X–XVIII) were successfully synthesized and screened for their acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) inhibitory activities. These molecules, most of which were synthesized for the first time (compounds V, XI–XV, XVII and XVIII are new, others are known), were completely characterized by elemental analysis and some spectroscopic methods such as mass spectroscopy, FT-IR, 1D NMR (1H- and 13C- NMR) and 2D NMR (COSY and HMQC). The results indicated that all tested molecules inhibited these enzymes at concentrations ranging from 80.4 to 247.1 μM. All tested molecules exhibited higher activities than the standard compound rivastigmine (IC50 = 501 ± 3.08 μM) against AChE. Among the synthesized molecules, the most active molecule was compound (XIII) (IC50 = 92.7 ± 0.9 μM) against AChE. The same molecules displayed lower activities than the standard compounds galanthamine (IC50 =7.96 ± 0.59 μM) and rivastigmine (IC50 = 19.95 ± 0.20 μM) against BChE. Also, physicochemical properties, pharmacokinetic properties, and drug-likeness of all tested molecules (I–XVIII) were calculated by using SwissADME.





Similar content being viewed by others
REFERENCES
Sathya, S., Manogari, B.G, Thamaraiselvi, K., Vaidevi, S., Ruckmani, K., and Devi K.P., Nutr. Neurosci., 2020, vol. 25, pp. 485–501. https://doi.org/10.1080/1028415X.2020.1764290
Çınar, E., Başaran, E., Erdoğan, Ö., Çakmak, R., Boğa, M., and Çevik, Ö., J. Chin. Chem. Soc., 2021, vol. 68, pp. 2355–2367. https://doi.org/10.1002/jccs.202100357
Yan, F., Tian, Y., Huang; Y., Wang, Q., Liu, P., Wang, N., Zhao, F., Zhong, L., Hui, W., and Luo, Y., Biomed. Pharmacother., 2022, vol. 145, p. 112453. https://doi.org/10.1016/j.biopha.2021.112453
Kaundal, R.K., Datusalia, A.K., and Sharma, S.S., Pharmacol. Res., 2022, vol. 175, p. 106018. https://doi.org/10.1016/j.phrs.2021.106018
Mirzaei, G. and Adeli, H., Biomed. Signal. Process. Control., 2022, vol. 72, p. 103293. https://doi.org/10.1016/j.bspc.2021.103293
Ju, Y. and Tam, K.Y., Neural Regener. Res., 2022, vol. 17, pp. 543–549. https://doi.org/10.4103/1673-5374.320970
Parihar, M.S. and Hemnani, T., J. Clin. Neurosci., 2004, vol. 11, pp. 456–467. https://doi.org/10.1016/j.jocn.2003.12.007
Tang, Y., Zhang, D., Gong, X., and Zheng, J., Biophys. Chem., 2020, vol. 281, p. 106735. https://doi.org/10.1016/j.bpc.2021.106735
Luo, J.E. and Li, Y.M., Cell Biosci., 2022, vol. 12, pp. 1–12. https://doi.org/10.1186/s13578-021-00738-7
Ruan, Z. Regen. Res., 2022, vol. 17, pp. 328–329. https://doi.org/10.4103/1673-5374.317975
Do Carmo, S., Kannel, B., and Cuello, A.C., Cell, 2022, vol. 11, pp. 1–15. https://doi.org/10.3390/cells11010016
Francis, P.T., Palmer, A.M., Snape, M., and Wilcock, G.K. J. Neurol. Neurosurg. Psychiatry, 1999, vol. 66, pp. 137–147. https://doi.org/10.1136/jnnp.66.2.137
Quinn, D.M., Chem. Rev., 1987, vol. 87, pp. 955–979. https://doi.org/10.1021/cr00081a005
Kawashima, K. and Fujii, T., Pharmacol. Ther., 2020, vol. 86, pp. 29–48. https://doi.org/10.1016/S0163-7258(99)00071-6
Topal, M., Int. J. Food Prop., 2019, vol. 22, pp. 1527–1535. https://doi.org/10.1080/10942912.2019.1656234
Santarpia, L., Grandone, I., Contaldo, F., and Pasanisi, F., J. Cachexia Sarcopenia Muscle, 2013, vol. 4, pp. 31–39. https://doi.org/10.1007/s13539-012-0083-5
Massoulie, J., Pezzementi, L., Bon, S., Krejci, E., and Valette, F.M., Prog. Neurobiol., 1993, vol. 41, pp. 31–91. https://doi.org/10.1016/0301-0082(93)90040-Y
Darvesh, S., Curr. Alzheimer Res., 2016, vol. 13, pp. 1173–1177. https://doi.org/10.2174/1567205013666160404120542
Summers, W.K., J. Alzheimer’s Dis., 2006, vol. 9, pp. 439–445. https://doi.org/10.3233/jad-2006-9s350
Crismon, M.L., Ann. Pharmacother., 1994, vol. 28, pp. 744–751. https://doi.org/10.1177/106002809402800612
Misson, J. and Kendall, M.J., J. Clin. Pharm. Ther., 1997, vol. 22, pp. 251–255. https://doi.org/10.1046/j.1365-2710.1997.10275102.x
Çakmak, R., Başaran, E., and Şentürk, M., Arch. Pharm., 2022, vol. 355, p. e2100430. https://doi.org/10.1002/ardp.202100430
Barner, E.L. and Gray, S.L., Ann. Pharmacother., 1998, vol. 32, pp. 70–77. https://doi.org/10.1345/aph.17150
Jann, M.W., Shirley, K.L., and Small, G.W., Pharmacokinet. Clin., 2002, vol. 41, pp. 719–739. https://doi.org/10.2165/00003088-200241100-00003
Prvulovic, D., Hampel, H., and Pantel, J., Expert Opin., Drug Metab. Toxicol., 2010, vol. 6, pp. 345–354. https://doi.org/10.1517/17425251003592137
Sramek, J.J., Frackiewicz, E.J., and Cutler, N.R., Expert Opin. Invest. Drugs, 2000, vol. 9, pp. 2393–2402. https://doi.org/10.1517/13543784.9.10.2393
Kumari, A. and Singh, R.K., Bioorg. Chem., 2019, vol. 89, p. 103021. https://doi.org/10.1016/j.bioorg.2019.103021
Sicak, Y., Oruç-Emre, E.E., Öztürk, M., Karaküçük-İyidoğan, A., and Nadeem, S., J. Heterocycl. Chem., 2020, vol. 57, pp. 830–841. https://doi.org/10.1002/jhet.3829
Chauhan, P., Mahajan, S., and Enders, D., Chem. Commun., 2015, vol. 51, p. 12890. https://doi.org/10.1039/C5CC04930J
Horton, D.A., Bourne, G.T., and Smythe, M.L., Chem. Rev., 2003, vol. 103, pp. 893–930. https://doi.org/10.1021/cr020033s
Brune, K., Acute Pain, 1997, vol. 1, pp. 33–40. https://doi.org/10.1016/S1366-0071(97)80033-2
Rogosch, T., Sinning, C., Podlewski, A., Watzer, B., Schlosburg, J., Lichtman, A.H., Cascio, M.G., Bisogno, T., Di Marzo, V., Nüsing, R., and Imming, P., Bioorg. Med. Chem., 2012, vol. 20, pp. 101–107. https://doi.org/10.1016/j.bmc.2011.11.028
Yoshida, H., Yanai, H., Namiki, Y., Fukatsu-Sasaki, K., Furutani, N., and Tada, N., CNS Drug Rev., 2006, vol. 12, pp. 9–20. https://doi.org/10.1111/j.1527-3458.2006.00009.x
Clark, M.P., Laughlin, S.K., Laufersweiler, M.J., Bookland, R.G., Brugel, T.A., Golebiowski, A., Sabat, M.P., Townes, J.A., VanRens, J.C., Djung, J.F., Natchus, M.G., De, B., Hsieh, L.C., Xu, S.C., Walter, R.L., Mekel, M.J., Heitmeyer, S.A., Brown, K.K., Juergens, K., Taiwo, Y.O., and Janusz, M.J., J. Med. Chem., 2004, vol. 47, pp. 2724–2727. https://doi.org/10.1021/jm049968m
Hadi, V., Koh, Y.H., Sanchez, T.W., Barrios, D., Neamati, N., and Jung, K.W., Bioorg. Med. Chem. Lett., 2010, vol. 20, pp. 6854–6857. https://doi.org/10.1016/j.bmcl.2010.08.057
Chande, M.S., Barve, P.A., and Suryanarayan, V., J. Heterocycl. Chem., 2007, vol. 44, pp. 49–53. https://doi.org/10.1002/jhet.5570440108
Alam, M.S., Choi, J.H., and Lee, D.U., Bioorg. Med. Chem., 2012, vol. 20, pp. 4103–4018. https://doi.org/10.1016/j.bmc.2012.04.058
Gürsoy, A., Demirayak, Ş., Çapan, G., Erol, K., and Vural, K., Eur. J. Med. Chem., 2000, vol. 35, pp. 359–364. https://doi.org/10.1016/S0223-5234(00)00117-3
Mohanram, I. and Meshram, J., Int. Sch. Res. Notices, 2014, vol. 2014, pp. 1–7. https://doi.org/10.1155/2014/639392
Rostom, S.A.F., El-Ashmawy, I.M., Abd El Razik, H.A., Badr, M.H., and Ashour, H.M.A., Bioorg. Med. Chem., 2009, vol. 17, pp. 882–895. https://doi.org/10.1016/j.bmc.2008.11.035
Ghorab, M.M., El-Gazzar, M.G., and Alsaid, M.S., Int. J. Mol. Sci., 2014, vol. 15, pp. 7539–7553. https://doi.org/10.3390/ijms15057539
Başaran, E., Inst. Sci. & Tech., 2021, vol. 11, pp. 2967–2978. https://doi.org/10.21597/jist.963129
Tok, F., Koçyiğit-Kaymakçıoğlu, B., Sağlık, B.N., Levent, S., Özkay, Y., and Kaplancıklı, Z.A., Bioorg. Chem., 2019, vol. 84, pp. 41–50. https://doi.org/10.1016/j.bioorg.2018.11.016
Shaikh, S., Dhavan, P., Singh, P., Uparkar, J., Vaidya, S.P., Jadhav, B.L., and Ramana M.M.V., J. Biomol. Struct. Dyn., 2021, pp. 1–16. https://doi.org/10.1080/07391102.2021.2006088
Yan, L. and Muller, C.E., J. Med. Chem., 2004, vol. 47, pp. 1031–1041. https://doi.org/10.1021/jm0310030
Zuse, A., Schmidt, P., Baasner, S., Böhm, K.J., Muller, K., Gerlach, M., Günther, E.G., Unger, E., and Prinz, H., J. Med. Chem., 2007, vol. 50, pp. 6059–6066. https://doi.org/10.1021/jm0708984
Hou. S., Yi, Y.W., Kang, H.J., Zhang, L., Kim, H.J., Kong, Y., Liu, Y., Wang, K., Kong, H.S., Grindrod, S., Bae, I., and Brown, M.L., J. Med. Chem., 2014, vol. 57, pp. 6342–6353. https://doi.org/10.1021/jm4018042
Carey, J.S., Laffan, D., Thomson. C., and Williams, M.T., Org. Biomol. Chem., 2006, vol. 4, pp. 2337–2347. https://doi.org/10.1039/B602413K
Elder, D., Facchine, K.L., Levy, J.N., Parsons, R., Ridge, D., Semo, L., and Teasdale, A., Org. Process Res. Dev., 2012, vol. 16, pp. 1707–1710. https://doi.org/10.1021/op300216x
Betts, L.M., Tam, N.C., Kabir, S.M.H., Langler, R.F., and Crandall, I., Aust. J. Chem., 2006, vol. 59, pp. 277–282. https://doi.org/10.1071/CH04299
Krátký, M., Štěpánková, Š., Vorčáková, K., Švarcová, M., and Vinšová, J., Molecules, 2016, vol. 21, pp. 191–201. https://doi.org/10.3390/molecules21020191
Krátký, M., Štěpánková, Š., Vorčáková, K., and Vinšová, J., Bioorg. Chem., 2015, vol. 58, pp. 48–52. https://doi.org/10.1016/j.bioorg.2014.11.005
Sicak, Y., Turk. J. Chem., 2022, vol. 46, pp. 665–676. https://doi.org/10.3906/kim-2107-27
Sicak, Y. Med. Chem. Res., 2021, vol. 30, pp. 1557–1568. https://doi.org/10.1007/s00044-021-02756-z
Tatar, G. and Kurşun Aktar, B.S., Int. J. Adv. Eng. Pure. Sci., 2021, vol. 33, pp. 660–669. https://doi.org/10.7240/jeps.945430
Chen, E.M., Lu, P.J., and Shaw, A.Y., J. Heterocycl. Chem., 2012, vol. 49, pp. 792–798. https://doi.org/10.1002/jhet.859
Zemplen, G. and Kisfaludy, L., Chem. Ber., 1960, vol. 93, pp. 1125–1128. https://doi.org/10.1002/cber.19600930521
Mahapatra, M.K., Kumar, R., and Kumar, M., Med. Chem. Res., 2018, vol. 27, pp. 476–487. https://doi.org/10.1007/s00044-017-2074-8
Julia, M., Manoury, P., and Voillaume, C., Bull. Soc. Chim. Fr., 1965, vol. 5, pp. 1411.
Zhang, Q.Z., Zhao, Y.L., Chen, X., and Yu, M., Acta Cryst., 2006, vol. E62, p. o5711. https://doi.org/10.1107/S1600536806048938
Mahapatra, M.K., Kumar, R., and Kumar, M., Bioorg. Chem., 2017, vol. 71, pp. 1–9. https://doi.org/10.1016/j.bioorg.2017.01.007
Olivera, R., SanMartin, R., Churruca, F., and Domínguez, E., J. Org. Chem., 2002, vol. 67, pp. 7215–7225. https://doi.org/10.1021/jo025767j
Chen, X. and Yu, M., Acta Cryst., 2007, vol. 63, p. o2936. https://doi.org/10.1107/S1600536806043546
Zhao, Y.L., Zhang, Q.Z., Chen. X., and Yu, M., Acta Cryst., 2006, vol. E62, p. o4590. https://doi.org/10.1107/S1600536806037950
Zilbeyaz, K., Stellenboom, N., Guney, M., Oztekin, A., and Senturk, M., J. Biochem. Mol. Toxicol., 2018, vol. 32, p. e22210. https://doi.org/10.1002/jbt.22210
http://www.swissadme.ch/. Accessed January 1, 2022.
ACKNOWLEDGMENTS
The author would like to thank Prof. Dr. Murat Şentürk (Department of Biochemistry of Faculty of Pharmacy of Ağrı İbrahim Çeçen University) for the laboratory support of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author declares that he has no conflicts of interest.
This article does not contain any studies involving animals or human participants performed by the author.
Supplementary Information
Rights and permissions
About this article
Cite this article
Eyüp Başaran Schiff Base Derivatives Based on Ampyrone as Promising Acetylcholinesterase Inhibitors: Synthesis, Spectral Characterization, Biological Activity, and SwissADME Predictions. Russ J Bioorg Chem 49, 114–126 (2023). https://doi.org/10.1134/S1068162023010065
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023010065