Abstract
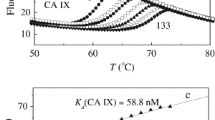
Sulfonamides is an important class of compounds, which can be used as carbonic anhydrase inhibitors. Nine different benzenesulfonamide compounds were synthesized, and their inhibitory effects on carbonic anhydrase II were studied by esterase method and molecular docking. The results showed that compounds (IId)–(IIg) with nitro and acetamide groups on the benzene ring exhibited excellent carbonic anhydrase II inhibitory activities. Molecular docking showed that compared with the control inhibitor acetazolamide, the compounds (IId)–(IIg) docked at the carbonic anhydrase II active site and showed higher binding energy and stronger binding ability. The physical and chemical properties of all compounds were studied by Molinspiration, which showed outstanding drug-like properties and ADME properties. Cytotoxicity assay results showed that compounds (IIe) and (IIf) were almost non-toxic to HepG2 and RAW264.7 cells. In conclusion, the compounds (IIe) and (IIf) have a certain application prospect as new inhibitors of carbonic anhydrase II.




Similar content being viewed by others
REFERENCES
Bayram, E., Senturk, M., Kufrevioglu, O.I., and Supuran, C.T., Bioorg. Med. Chem., 2008, vol. 16, pp. 9101–9105. https://doi.org/10.1016/j.bmc.2008.09.028
Bal, S., Kaya, R., Gök, Y., Taslimi, P., Aktaş, A., Karaman, M., and Gülçin, I., Bioorg. Chem., 2019, vol. 23, p. 103 468. https://doi.org/10.1016/j.bioorg.2019.103468
Mann, T. and Keilin, D., Nature, 1940, vol. 146, pp. 164–165. https://doi.org/10.1038/146164a0
Serbian, I., Schwarzenberger, P., Loesche, A., Hoenke, S., Al-Harrasi, A., and Csuk, R., Bioorg. Chem., 2019, vol. 91, p. 103 123. https://doi.org/10.1016/j.bioorg.2019.103123
Ahmed, A., Channar, P.A., Saeed, A., Kalesse, M., Kazi, M.A., Larik, F.A., Abbas, Q., Hassan, M., Raza, H., and Seo, S.Y.J., Bioorg. Chem., 2019, vol. 86, pp. 624–630. https://doi.org/10.1016/j.bioorg.2019.01.060
EntezariHeravi, Y., Sereshti, H., Saboury, A.A., Ghasemi, J., Amirmostofian, M., and Supuran, C.T.J., Enzyme. Inhib. Med. Chem., 2017, vol. 32, pp. 688–700. https://doi.org/10.1080/14756366.2016.1241781
Vats, L., Kumar, R., Bua, S., Nocentini, A., Gratteri, P., Supuran, C.T., and Sharma, P.K., Eur. J. Med. Chem., 2019, vol. 183, p. 111 698. https://doi.org/10.1016/j.ejmech.2019.111698
Akin, S., Ayaloglu, H., Gultekin, E., Colak, A., Bekircan, O., and Akatin, M.Y., Bioorg. Chem., 2019, vol. 83, pp. 170–179. https://doi.org/10.1016/j.bioorg.2018.10.042
Masini, E., Carta, F., Scozzafava, A., and Supuran, C.T., Expert. Opin. Ther. Pat., 2013, vol. 23, pp. 705–716. https://doi.org/10.1517/13543776.2013.794788
Scozzafava, A. and Supuran, C.T., Subcell. Biochem., 2013, vol. 75, pp. 349– 359. https://doi.org/10.1007/978-94-007-7359-2_17
Wang, Y., Guo, H., Tang, G., He, Q., Zhang, Y., Hu, Y.,Wang, Y., and Lin, Z., Comput. Biol. Chem., 2019, vol. 80, pp. 234–243. https://doi.org/10.1016/j.compbiolchem.2019.03.005
Supuran, C.T., Briganti, F., Tilli, S., Chegwidden, W.R., and Scozzafava, A., Bioorg. Med. Chem., 2001, vol. 9, pp. 703–714. https://doi.org/10.1016/S0968-0896(00)00288-1
Innocenti, A., Vullo, D., Scozzafava, A., and Supuran, C.T., Bioorg. Med. Chem. Lett., 2008, vol. 18, pp. 1583–1587. https://doi.org/10.1016/j.bmcl.2008.01.077
Innocenti, A., Beyza Öztürk Sarıkaya, S., Gülçin, İ., and Supuran, C.T., Bioorg. Med. Chem., 2010, vol. 18, pp. 2159–2164. https://doi.org/10.1016/j.bmc.2010.01.076
Supuran, C.T. and Claudiu, T., Future. Med. Chem., 2011, vol. 3, pp. 1165–1180. https://doi.org/10.4155/fmc.11.69
Küçükbay, H., Buğday, N., Küçükbay, F.Z., Berrino, E., Bartolucci, G., Del Prete, S., Capasso, C., and Supuran, C.T., Bioorg. Chem., 2019, vol. 83, pp. 414–423. https://doi.org/10.1016/j.bioorg.2018.11.003
Ghorab, M.M., Alsaid, M.S., Ceruso, M., Nissan, Y.M., and Supuran, C.T. Bioorg. Med. Chem., 2014, vol. 22, pp. 3684–3695. https://doi.org/10.1016/j.bmc.2014.05.009
Alaei, L., Khodarahmi, R., Sheikh-Hasani, V., Sheibani, N., and Moosavi-Movahedi, A.A., Int. J. Biol. Macromol., 2018, vol. 120, pp. 1198–1207. https://doi.org/10.1016/j.ijbiomac.2018.08.186
Shankara, S., Pangeni, R., Park, J.W., and Rhim, J.W., Mater. Sci. Eng. C., 2018, vol. 92, pp. 508–517. https://doi.org/10.1016/j.msec.2018.07.015
Husain, A., Ahmad, A., Khan, S.A., Asif, M., Bhutani, R., and Al-Abbasi, F.A., Saudi. Pharm. J., 2016, vol. 24, pp. 104–114. https://doi.org/10.1016/j.jsps.2015.02.008
Funding
This work was supported by the National Natural Science Foundation of China (no. 31760521).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving human participants performed by any of the authors and does not contain any studies involving animals performed by any of the authors.
Conflict of Interests
The authors state that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, A.M., Wei, N., Liu, X.F. et al. Inhibitory Evaluation and Molecular Docking Analysis of Benzenesulfonamides on Carbonic Anhydrase II. Russ J Bioorg Chem 47, 261–269 (2021). https://doi.org/10.1134/S1068162021010283
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162021010283