Abstract
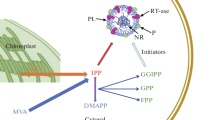
Natural rubber, cis-1,4-polyisoprene, is an essential raw material used in thousands of products, many of which are absolutely necessary for medical purposes. Natural rubber is obtained from latex, an aqueous emulsion present in the laticiferous vessels of the natural rubber-producing plants. Hevea brasiliensis (the Brazilian rubber tree) currently is the only commercially important source of natural rubber. H. brasiliensis crops have very little genetic variability, leaving rubber plantations at risk of serious pathogenic attacks. In addition, repeated exposure to residual proteins in latex products derived from H. brasiliensis have led to serious and widespread allergic (type I) hypersensitivity. Therefore, identification of alternative sources of natural rubber is a very important biotechnological task. Potentially, Russian dandelion (Taraxacum kok-saghyz) may be such an alternative because significant amounts of natural rubber are produced in its root system. However, H. brasiliensis is a more efficient producer of natural rubber than T. kok-saghyz. Thus, improvement of rubber biosynthesis in plants is a first-priority problem of modern biotechnology. In this review, we describe proteins that may increase the concentration of natural rubber in laticiferous vessels of T. kok-saghyz and its close relative Taraxacum brevicorniculatum, when overexpressed in the plants. These proteins, cis-prenyltransferases, rubber transferase activator, and small rubber particle proteins, are directly involved in synthesis of the polyisoprene chain. We also analyze the effects of their expression levels on the production of natural rubber in vivo.
Similar content being viewed by others
Abbreviations
- Ac-CоА:
-
acetyl coenzyme A
- APP:
-
allylpyrophosphate
- CPT1–3:
-
cis-prenyltransferases 1–3
- DMAPP:
-
1,1-dimethylallylpyrophosphate
- FPP:
-
farnesyl pyrophosphate
- HMG-CoA:
-
3-hydroxy-3-methylglutaryl coenzyme A
- IPP:
-
iso-pentenylpyrophosphate
- HRT1 and HRT2:
-
cis-prenyltransferases 1 and 2
- NgBR:
-
Nogo-B receptor
- TbRTA:
-
T. brevicorniculatum cis-prenyltransferase activator
- RNAi:
-
RNA interference
- SRPP:
-
small rubber particle-associated protein
- EPR:
-
endoplasmic reticulum
References
Eng A. H., Ong E. L. Hevea natural rubber, in Plastics Engineering: Handbook of Elastomers, Bhowmick, A.K. and Stephens, H.L., Eds., New York: Marcel Dekker, 2000, vol. 61, pp. 29–59.
McIntyre, D., Stephens, H.L., Schloman, W.W., Jr., and Bhowmick, A.K., Guayule rubber, in Plastics Engineering: Handbook of Elastomers, Bhowmick, A.K. and Stephens, H.L., Eds., New York: Marcel Dekker, 2000, vol. 61, pp. 1–27.
Puskas, J.E., Producers and world market of synthetic rubbers, in Biopolymers, Polyisoprenoids, Koyama, E. and Steinbuchel, A., Eds., Weinheim: Wiley, 2001, vol. 2, pp. 287–320.
Cornish, K., Hypoallergenic natural rubber products from Parthenium argentatum (gray) and other non-Hevea brasiliensis species, US Patent nos. 5580942, 1996; 5717050, 1998.
Nyburg, S.C., Acta Crystallogr., 1954, vol. 7, pp. 385–392.
Tanaka, Y., Prog. Polym. Sci., 1989, vol. 14, pp. 339–371.
Swanson, C.L., Buchana, R.A., and Otey, F.H., J. Appl. Polym. Sci., 1979, vol. 23, pp. 743–748.
Castillon, J. and Cornish, K., Phytochemistry, 1999, vol. 51, pp. 43–51.
Cornish, K., Castillon, J., and Scott, D.J., Biomacromolecules, 2000, vol. 1, pp. 632–641.
Ownby, D.R., Ownby, H.E., McCullough, J., and Shafer, A.W., J. Allergy Clin. Immunol., 1996, vol. 97, pp. 1188–1192.
Pailhories, G., Clin. Rev. Allergy, 1993, vol. 11, pp. 391–402.
Tomazic, V.J., Withrow, T.J., Fisher, B.R., and Dillard, S.F., Clin. Immunol. Immunopathol., 1992, vol. 64, pp. 89–97.
Morales, C., Basomba, A., Carreira, J., and Sastre, A., Clin. Exp. Allergy, 1989, vol. 19, pp. 425–430.
Slater, J.E., N. Engl. J. Med., 1989, vol. 320, pp. 1126–1130.
Mooibroek, H. and Cornish, K., Appl. Microbiol. Biotechnol., 2000, vol. 53, pp. 355–365.
Tanaka, Y., Aik-Hwee, E., Ohya, N., Nishiyama, N., Tangpakdee, J., Kawahara, S., and Wititsuwannakul, R., Phytochemistry, 1996, vol. 41, pp. 1501–1505.
Cornish, K., Eur. J. Biochem., 1993, vol. 218, pp. 267–271.
Cornish, K. and Backhaus, R.A., Phytochemistry, 1990, vol. 29, pp. 3809–3813.
Madhavan, S., Greenblatt, G.A., Foster, M.A., and Benedictc, R., Plant Physiol., 1989, vol. 89, pp. 506–511.
Archer, B.L., Audley, B.G., Cockbain, E.G., and McSweeney, G.P., Biochem. J., 1963, vol. 89, pp. 565–574.
Cornish, K. and Siler, D.J., Plant Physiol. Biochem., 1996, vol. 34, pp. 377–384.
Chappell, J., Annu. Rev. Plant Physiol. Plant Mol. Biol., 1995, vol. 46, pp. 521–547.
Holstein, S.A. and Hohl, R.J., Lipids, 2004, vol. 39, pp. 293–309.
Goldstein, J.L. and Brown, S.B., Nature, 1990, vol. 343, pp. 425–430.
Miziorko, H., Arch. Biochem. Biophys., 2011, vol. 505, pp. 131–143.
Dellas, N., Thomas, S.T., Manning, G., and Noel, J.P., eLife, 2013, vol. 2, e00672.
Vinokur, J.M., Korman, T.P., Cao, Z., and Bowie, J.U., Biochemistry, 2014, vol. 53, pp. 4161–4168.
Azami, Y., Hattori, A., Nishimura, H., Kawaide, H., Yoshimura, T., and Hemmi, H., J. Biol. Chem., 2014, vol. 289, pp. 15957–15967.
Seetang-Nun, Y., Sharkey, T.D., and Suvachittanont, W., J. Plant Physiol., 2008, vol. 165, pp. 991–1002.
Sando, T., Takeno, S., Watanabe, N., Okumoto, H., Kuzuyama, T., Yamashita, A., Hattori, M., Ogasawara, N., Fukusaki, E., and Kobayashi, A., Biosci. Biotechnol. Biochem., 2008, vol. 72, pp. 2903–2917.
Archer, B.L. and Audley, B.G., Linn. Soc., 1987, vol. 94, pp. 181–196.
Scott, D.J., Da Costa, B.M.T., Espy, S.C., Keasling, J.D., and Cornish, K., Phytochemistry, 2003, vol. 64, pp. 123–134.
Puskas, J.E., Gautriaud, E., Deffieux, A., and Kennedy, J.P., Prog. Polym. Sci., 2006, vol. 31, pp. 533–548.
Yokozawa, T. and Yokoyama, A., Chem. Rec., 2005, vol. 5, pp. 47–57.
Yokozawa, T. and Yokoyama, A., Polym. J., 2004, vol. 36, pp. 65–83.
Liang, P.H., Ko, T.-P., and Wang, A.H.J., Eur. J. Biochem., 2002, vol. 269, pp. 3339–3354.
Arias, M., Hernandez, M., Remondegui, N., Huvenaars, K., van Dijk, P., and Ritter, E., Sci. Rep., 2016, vol. 6, p. 31031.
Zhang, Y., Iaffaldano, B.J., Zhuang, X., Cardina, J., and Cornish, K., BMC Plant Biol., 2017, vol. 17, pp. 1–34.
van Beilen, J.B. and Poirier, Y.., Crit. Rev. Biotech., vol. 27, pp. 217–231.
Cornish, K., Phytochemistry, 2001, vol. 57, pp. 1123–1134.
Siler, D.J. and Cornish, K., Phytochemistry, 1993, vol. 32, pp. 1097–1102.
Takahashi, S. and Koyama, T., Chem. Rec., 2006, vol. 6, pp. 194–205.
Bouvier, F., Rahier, A., and Camara, B., Prog. Lipid. Res., 2005, vol. 44, pp. 357–429.
Lange, B.M. and Ghassemian, M., Plant. Mol. Biol., 2003, vol. 51, pp. 925–948.
Cunillera, N., Arró, M., Delourme, D., Karst, F., Boronat, A., and Ferrer, A., J. Biol. Chem., 1996, vol. 271, pp. 7774–7780.
Oh, S.K., Han, K.H., Ryu, S.B., and Kang, H., J. Biol. Chem., 2000, vol. 275, pp. 18482–18488.
Asawatreratanakul, K., Zhang, Y.W., Wititsuwannakul, D., Wititsuwannakul, R., Takahashi, S., Rattanapittayaporn, A., and Koyama, T., Eur. J. Biochem., 2003, vol. 270, pp. 4671–4680.
Schmidt, T., Lenders, M., Hillebrand, A., van Deenen, N., Munt, O., Reichelt, R., Eisenreich, W., Fischer, R., Prufer, D., and Gronover, C.S., BMC. Biochem., 2010, vol. 11, p. 11.
Schmidt, T., Hillebrand, A., Wurbs, D., Wahler, D., Lenders, M., Schulze Gronover, C., and Prufer, D., Plant Mol. Biol. Rep., 2010, vol. 28, pp. 277–284.
Post, J., van Deenen, N., Fricke, J., Kowalski, N., Wurbs, D., Schaller, H., Eisenreich, W., Huber, C., Twyman, R.M., Prufer, D., and Schulze Gronover, C., Plant Physiol., 2012, vol. 158, pp. 1406–1417.
Harrison, K.D., Park, E.J., Gao, N., Kuo, A., Rush, J.S., Waechter, C.J., Lehrman, M.A., and Sessa, W.C., EMBO J., 2011, vol. 30, pp. 2490–2500.
Kharel, Y. and Koyama, T., Nature Prod. Rep., 2003, vol. 20, pp. 111–118.
Epping, J., van Deenen, N., Niephaus, E., Stolze, A., Fricke, J., Huber, C., Eisenreich, W., Twyman, R., Prufer, D., and Gronover, C.S., Nature Plants, 2015, vol. 1, p. 15048.
Park, E.J., Grabinska, K.A., Guan, Z., Stranecky, V., Hartmannova, H., Hodanova, K., Baresova, V., Sovova, J., Jozsef, L., Ondruskova, N., Hansikova, H., Honzik, T., Zeman, J., Hulkova, H., Wen, R., Kmoch, S., and Sessa, W.C., Cell Metab., 2014, vol. 20, pp. 448–457.
Grabinska, K.A., Cui, J., Chatterjee, A., Guan, Z., Raetz, C.R., Robbins, P.W., and Samuelson, J., Glycobiology, 2010, vol. 20, pp. 824–832.
Dennis, M.S. and Light, D.R., J. Biol. Chem., 1989, vol. 264, pp. 18608–18617.
Kush, A., Goyvaerts, E., Chye, M.L., and Chua, N.H., Proc. Natl. Acad. Sci. U. S. A., 1990, vol. 87, pp. 1787–1790.
Han, K.H., Shin, D.H., Yang, J., Kim, I.J., Oh, S.K., and Chow, K.S., Tree Physiol., 2000, vol. 20, pp. 503–510.
Chow, K.S., Wan, K.L., Isa, M.N., Bahari, A., Tan, S.H., Harikrishna, K., and Yeang, H.Y., J. Exp. Bot., 2007, vol. 58, pp. 2429–2440.
Oh, S.K., Kang, H., Shin, D.H., Yang, J., Chow, K.S., Yeang, H.Y., Wagner, B., Breiteneder, H., and Han, K.H., J. Biol. Chem., 1999, vol. 274, pp. 17132–17138.
Yeang, H.Y., Ward, M.A., Zamri, A.S., Dennis, M.S., and Light, D.R., Allergy, 1998, vol. 53, pp. 513–519.
Wahler, D., Gronover, C.S., Richter, C., Foucu, F., Twyman, R.M., Moerschbacher, B.M., Fischer, R., Muth, J., and Prufer, D., Plant Physiol., 2009, vol. 151, pp. 334–346.
Schmidt, T., Hillebrand, A., Wurbs, D., Wahler, D., Lenders, M., Schulze Gronover, C., and Prufer, D., Plant Mol. Biol. Rep., 2009, vol. 28, pp. 277–284.
Collins-Silva, J., Taban Nural, A., Skaggs, A., Scott, D., Hathwaik, U., Woolsey, R., Schegg K., McMahan, C., Whalen, M., Cornish, K., and Shintani, D., Phytochemistry, 2012, vol. 79, pp. 46–56.
van den Ende, W., Michiels, A., van Wonterghem, D., Vergauwen, R., and van Laere, A., Plant Physiol., 2000, vol. 123, pp. 71–80.
De Roover, J., van Laere, A., De Winter, M., Timmermans, J.W., and van den Ende, W., Physiol. Plant., 1999, vol. 106, pp. 28–34.
Stolze, A., Wanke, A., van Deenen, N., Geyer, R., Prufer, D., and Gronover, C.S., Plant Biotechnol. J, 2017, vol. 15, pp. 740–753.
Chappell, J., Wolf, F., Proulx, J., Cuellar, R., and Saunders, C., Plant Physiol., 1995, vol. 109, pp. 133–1343.
Basson, M.E., Thorsness, M., and Rine, J., Proc. Natl. Acad. Sci. U. S. A., 1986, vol. 83, p. 5563.
Conolly, J.D. and Hill, R.A., Dictionary of Terpenoids, New York: Chapman & Hall, 1992.
Woitek, S., Unkles, S.E., Kinghorn, J.R., and Tudzynski, B., Curr. Genet., 1997, vol. 31, pp. 38–47.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.Yu. Amerik, Yu.Tc. Martirosyan, I.V. Gachok, 2018, published in Bioorganicheskaya Khimiya, 2018, Vol. 44, No. 2, pp. 126–137.
Rights and permissions
About this article
Cite this article
Amerik, A.Y., Martirosyan, Y.T. & Gachok, I.V. Regulation of Natural Rubber Biosynthesis by Proteins Associated with Rubber Particles. Russ J Bioorg Chem 44, 140–149 (2018). https://doi.org/10.1134/S106816201801003X
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106816201801003X