Abstract
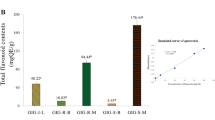
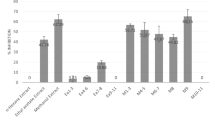
Accessible triterpenoids of ursane and lupane series, the flavonoid dihydroquercetin and their synthetic derivatives with polar substituents were tested in vitro for inhibition of collagenase 1 (MMP-1) in UVB irradiation assay. Ursolic acid and uvaol disuccinate were the most active inhibitors in the ursane series. In the lupane series, the best inhibition was manifested by carboxymethyl ester of betulonic acid and betulin succinates. Down- regulation of MMP-1 by dihydroquercetin and its synthetic derivatives surpassed the activity of a standard (retinoic acid).
Similar content being viewed by others
Abbreviations
- MMP-1:
-
matrix metalloproteinase-1; collagenase-1
- IL-8:
-
Interleukin-8
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- FBS:
-
fetal bovine serum
- PBS:
-
phosphate buffered saline
- HaCaT cells:
-
Immortalized human Keratinocytes
References
Vincenti, M.P., White, L.A., Schroen, D.J., Benbow, U., and Brinckerhoff, C.E., Crit. Rev. Eukaryot. Gene Expr., 1996, vol.6, pp. 391–411.
Fisher, G.J., Choi H.C., Bata-Csorgo, Z., Shao, Y., Datta, S., Wang, Z.Q., Kang, S., and Voorhees, J.J., J. Invest. Dermatol., 2001, vol. 117, pp. 219–226.
Chambers, A.F. and Matrisian, L.M., J. Natl. Cancer Inst., 1997, vol. 89, 1260–7120.
Zhou, L.Z., Johnson, A.P., and Rando, T.A., Free Rad. Biol. Med., 2001, vol. 31, pp.1405–1416.
Onoue, S., Kobayashi, T., Takemoto, Y., Sasaki, I., and Shinkai, H., J. Dermatol. Sci., 2003, vol. 33, pp. 105–111.
Varani, J., Warner, R.L., Gharaee-Kermani, M., Phan, S.H., Kang, S., Chung, J.H., Wang, Z.Q., Datta, S.C., Fisher, G.J., and Voorhees, J.J., J. Invest. Dermatol., 2000, vol. 114, pp. 480–486.
Kligman, A.M., Grove, G.L., Hirose, R., and Leyden, J.J., J. Am. Acad. Dermatol., 1986, vol. 15, pp. 836–859.
Lateef, H., Stevens, M.J., and Varani, J., Am. J. Pathol., 2004, vol. 165, pp. 167–174.
Moon, H.-I., Chung, J.H., Lee, J.-K., and Zee, O.-P., Arch. Pharm. Res., 2004, vol. 27, pp. 730–733.
Mix, K.S., Mengshol, J.A., Benbow, U., Vincenti, M.P., Sporn, M.B., and Brinckerhoff, C.E., Arthritis Rheum., 2001, vol. 44, pp. 1096–1104.
Cha, H.J., Park, M.T., and Chung, H.Y., Oncogene, 1998, vol. 6, pp. 771–778.
Es-Saady, D., Simon, A., Jayat-Vignoles, C., et al., Anticancer Res., 1996, vol. 16, pp. 481–486.
Han, S.K., Yi, K.O., and Park, S.J., Lipids, 1997, vol. 32, pp. 769–773.
Lin, W.-Y., Sadhasivam, S., and Lin, F.-H., Proc. Biochem., 2009, vol. 44, no. 6, pp. 678–684.
Sun, H.H., Kaplita, P.V., Houck, D.R., Stawicki, M.B., McGarry, R., Wahl, R.C., Gillum, A., and Cooper, R., Phytother. Res., 1996, vol. 10, pp. 194–197.
Zhang, Y.Wu., Ren, Y., Fu, J., and Zhang, Y., Food Chem. Toxicol., 2004, vol. 42, pp. 1867–1875.
Chaturvedula, V.S.P., Schilling, J.K., Johnson, R.K., and Kingston, D.G.I., J. Nat. Prod., 2003, vol. 66, pp. 419–422.
Lee, C.-W., Choi, H.-J., Kim, H.-S., Kim, D.-H., Chang, I.-S., Moon, H.T., Lee, S.-Y., Oh, W.K., and Woo, E.-R., Bioorg. Med. Chem., 2008, vol. 16, pp. 732–738.
Kuhareva, T.S., Krasnov, V.A., Koroteev, M.P., Kaziev, G.Z., Kuleshova, L.N., Korlyukov, A.A., Antipin, M.Yu., and Nifantiev, E.E., Russ. J. Org. Chem., 2004, vol. 40, pp. 1237–1240.
Vlahopoulos, S., Boldogh, I., Casola, A., and Brasier, A.R., Blood, 1999, vol. 94, pp. 1878–1889.
Sang, S., Lapsley, K., Rosen, R.T., and Ho, C.-T., J. Agric. Food Chem., 2002, vol. 50, pp. 607–609.
Xu, H.-X., Sim, K.-Ye., Zeng, F.-Q., and Wan, M., US Patent No. 5916919, Chem. Abstr., vol. 131, p. 80.
Tkachev, A.V. and Denisov, A.Yu., Tetrahedron, 1994, vol. 50, pp. 2591–2598.
Ryabinin, A.A., Zh. Obsch. Khim., 1946, vol. 16, pp. 771–774.
Kashiwada, Y., Nagao, T., Hashimoto, A., Ikeshiro, Y., Okabe, H., Cosentino, L. M., and Lee, K.-H., J. Nat. Prod., 2000, vol. 63, pp. 1619–1622.
Lee, E.H., Popov, S.A., Lee, J.Y., Shpatov, A.V., Kukina, T.P., Kang, S. W., Pan, C.-H., Um, B.H., and Jung, S.H., Russ. J. Bioorg. Chem., 2012 (in press).
Collins, D.O., Ruddock, P.L.D., Chiverton de Grasse, J., Reynolds, W.F., and Reese, P.B., Phytochemistry, 2002, vol. 59, pp. 479–488.
Hayek, E.W.H., Jordis, U., Moche, W., and Sauter, F., Phytochemistry, 1989, vol. 28, pp. 2229–2239.
Krasutsky, P.A., Carlson, R.M., and Karim, R., US Patent No. 6642217, 2003.
Sun, I.C., Wang, H.-K., Kashiwada, Y., Shen, J.-K., Cosentino, L. M., Chen, C.-H., Yang, L.-M., and Lee, K.-H., J. Med. Chem., 1998, vol. 41, pp. 4648–4657.
Petrenko, N.I., Elantseva, N.V., Petukhova, V.Z., Shakirov, M.M., Shults, E.E., and Tolstikov, G.A., Chem. Nat. Comp., 2002, vol. 38, pp. 331–339.
Nakagawa, H., Takaishi, Yo., Fujimoto, Yo., Duque, C., Garzon, C., Sato, M., Okamoto, M., Oshikawa, T., and Ahmed, Sh.U., J. Nat. Prod., 2004, vol. 67, pp. 1919–1924.
Barthel, A., Stark, S., and Csuk, R., Tetrahedron, 2008, vol. 64, pp. 9225–9229.
Zimmerman, D.W., J. Educat. Behav. Stat., 1997, vol. 22, pp. 349–360.
Author information
Authors and Affiliations
Corresponding authors
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Lee, C.W., Park, N.H., Kim, J.W. et al. Study of skin anti-ageing and anti-inflammatory effects of dihydroquercetin, natural triterpenoinds, and their synthetic derivatives. Russ J Bioorg Chem 38, 328–334 (2012). https://doi.org/10.1134/S1068162012030028
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162012030028