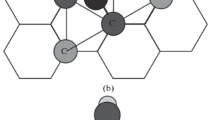
Abstract
By means of quantum chemistry, it is established that tungsten and rhenium are most effective alloying elements capable of increasing the resistance of aluminum alloys to hydrogen-induced embrittlement. Tungsten (W) and rhenium (Re) atoms produce strong compression of aluminum lattice, while having sufficiently large covalent radii. In addition, each W and Re atom forms stable chemical bonds with 12 surrounding Al atoms. As a result, W and Re dopants strongly bind Al atoms, thus significantly increasing the energy of vacancy formation and suppressing the process of hydrogen-induced embrittlement. The main mechanical properties of the most hydrogen-resistant aluminum compound, WReAl24, have been calculated using the density functional theory.



Similar content being viewed by others
REFERENCES
Aluminium Alloys—Recent Trends in Processing, Characterization, Mechanical Behavior and Applications, Ed. by S. Sivasankaran (InTech, Rijeka, 2017). https://doi.org/10.5772/68032
L. Soler, J. Macanas, M. Munoz, and J. Casado, J. Power Sources 169, 144 (2007). https://doi.org/10.1016/j.jpowsour.2007.01.080
J. B. von Colbe, J.-R. Ares, J. Barale, M. Baricco, C. Buckley, G. Capurso, N. Gallat, D. M. Grant, M. N. Guzik, I. Jacob, E. H. Jensen, T. Jensen, J. Jepsen, T. Klassen, M. V. Lototskyy, et al., Int. J. Hydrogen Energy 44, 7780 (2019). https://doi.org/10.1016/j.ijhydene.2019.01.104
M. Nagumo, Fundamental of Hydrogen Embrittlement (Springer, Singapore, 2016). https://doi.org/10.1007/978-981-10-0161-1
J. R. Scully, G. A. Young, and S. W. Smith, in Gaseous Hydrogen Embrittlement of Materials in Energy Technologies, Ed. by R. P. Gangloff and B. P. Somerday (Woodhead, Oxford, 2012), Vol. 1, p. 707.
D. A. Indeitsev and E. V. Osipova, Tech. Phys. Lett. 45, 882 (2019). https://doi.org/10.1134/S1063785019090074
R. V. Regel’, A. I. Slutsker, and E. E. Tomashevskii, The Kinetic Nature of the Strength of Solids (Nauka, Moscow, 1974) [in Russian].
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996). https://doi.org/10.1103/PhysRevLett.77.3865
B. Cordero, V. Gomez, A. E. Platero-Prats, M. Reves, J. Echeverria, E. Cremades, F. Barragan, and S. Alvarez, Dalton Trans., No. 21, 2832 (2008). https://doi.org/10.1039/B801115J
V. I. Kolesnikov, Yu. F. Migal, I. V. Kolesnikov, and E. S. Novikov, J. Friction Wear 36, 1 (2015). https://doi.org/10.3103/S1068366615010055
G. Kresse and J. Furthmuller, Phys. Rev. B 54, 11169 (1996). https://doi.org/10.1103/PhysRevB.54.11169
L. F. Mondolfo, Aluminum Alloys: Structure and Properties (Butterworth, London, 1976).
D. A. Indeitsev and E. V. Osipova, Phys. Solid State 51, 1901 (2009).
D. A. Indeitsev and E. V. Osipova, Dokl. Phys. 56, 523 (2011).
G. Simmons and H. Wang, Single Crystal Elastic Constants and Calculated Aggregate Properties, A Handbook (MIT Press, Cambridge, 1971).
D. A. Indeitsev and E. V. Osipova, Dokl. Phys. Chem. 484, 4 (2019). https://doi.org/10.31857/S0869-5652484156-60
Funding
This work was performed in the framework of a state assignment to the Institute of Problems of Mechanical Engineering (Russian Academy of Sciences, St. Petersburg), project no. AAAA-A18-118012790011-3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated by P. Pozdeev
Rights and permissions
About this article
Cite this article
Indeitsev, D.A., Osipova, E.V. An Analysis of the Hydrogen Embrittlement Resistance of Aluminum Alloys. Tech. Phys. Lett. 47, 170–173 (2021). https://doi.org/10.1134/S1063785021020231
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063785021020231