Abstract
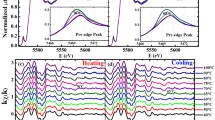
It is shown that an excess of free electrons occurs in hydrogenated films of vanadium sesquioxide compared to non-hydrogenated films. This leads to a shift of the temperature hysteresis loops of the insulator-metal thermal phase transition to the low-temperature region, i.e., to a decrease in the critical temperature Tc of the phase transition. The interpretation of the results is based on the fact that V2O3 is a highly correlated material, and the phase transition in it has a complex character: the Mott electronic transition initiates the Peierls structural transition. It is shown that the atoms of embedded hydrogen are localized on oxygen ions forming additional σ-bonds with them, the thermal destruction of some of which leads to an increase in the concentration of free electrons in conduction band of hydrogenated V2O3 films.




Similar content being viewed by others
REFERENCES
D. Wegkamp and J. Stáhler, Prog. Surf. Sci. 90, 464 (2015).
T. Lin, J. Wang, G. Liu, L. Wang, X. Wang, and Y. Zhang, Materials 10, 633 (2017). https://doi.org/10.3390/ma10060633
A. Tselev, A. Kolmakov, N. Lavrik, and S. Kalinin, Adv. Funct. Mater. 23, 2635 (2013).
A. V. Il’inskii, R. A. Kastro, M. E. Pashkevich, and E. B. Shadrin, Semiconductors 54, 205 (2020).
A. V. Il’inskii, O. E. Kvashenkina, and E. B. Shadrin, Semiconductors 45, 1153 (2011).
A. V. Il’inskii, O. E. Kvashenkina, and E. B. Shadrin, Semiconductors 46, 422 (2012).
A. V. Il’inskii and E. B. Shadrin, Phys. Solid State 62, 1451 (2020).
H. Ji, Thesis (Houston, TX, 2015).
H. Griesser, R. Öhrlein, W. Schwab, R. Ehrler, and V. Jáger, Org. Synth. 77, 236 (2000). https://doi.org/10.15227/077.0236
G. Ryder, World Social Protection Report (2018). www.ilo.org/dyn/icsc/.
T. B. Phan, M. Breugst, and H. Mayr, Angew. Chem. Int. Ed. 45, 3869 (2006). https://doi.org/10.1002/anie.200600542
R. A. Aliev, V. N. Andreev, V. M. Kapralova, V. A. Klimov, A. I. Sobolev, and E. B. Shadrin, Phys. Solid State 48, 929 (2006).
I. S. Gibin and P. E. Kotlyar, Prikl. Fiz., No. 5, 44 (2019).
K. Schneider, J. Mater. Sci.: Mater. Electron. 31, 10478 (2020). https://doi.org/10.1007/s10854-020-03596-0
A. Zylbersztein and N. F. Mott, Phys. Rev. B 11, 4383 (1975). https://doi.org/10.1103/PhysRevB.11.4383
V. Vonsovsky and M. I. Katsnelson, Quantum Solid-State Physics (Nauka, Moscow, 1983; Springer, Berlin, 1989).
T. F. Fuerst, E. P. Petsalis, S. T. B. Lundin, J. Wilcox, J. Douglas, W. Colin, and A. Wolden, J. Phys. Chem. 122, 3488 (2018).
G. G. Khaidarov, A. G. Khaidarov, and A. Ch. Mashek, Vestn. SPb. Univ., Ser. 4 (1), 3 (2011).
L. W. Barr, in Diffusion in Materials, Proceedings of the DIMAT 96 Conference, Ed. by H. Mehrer, Chr. Herzig, N. A. Stolwijk, and H. Bracht (Scitec Publ., 1997), Vol. 1, p. 1.
L. D. Landau and E. M. Lifshitz, Course of Theoretical Physics, Vol. 9: Statistical Physics, Part 2 (Fizmatlit, Moscow, 2004; Pergamon, New York, 1980).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by S. Rostovtseva
Rights and permissions
About this article
Cite this article
Il’inskii, A.V., Nikulin, E.I. & Shadrin, E.B. Mechanism of Hydrogen Effect on the Phase Transition in V2O3 Films. Phys. Solid State 63, 714–721 (2021). https://doi.org/10.1134/S1063783421050097
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063783421050097