Abstract
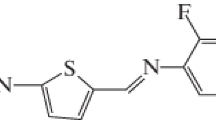
A novel Schiff base compound was synthesized and characterized by IR spectroscopy and X-ray diffraction method. The compound crystallizes in monoclinic space group P21/c with Z = 4 in the unit cell. The ring systems are perfectly planar but the whole molecule is not planar. The dihedral angle between the thiophene and phenyl rings is 38.7(3)°. In the crystal, intermolecular C–H⋅⋅⋅O interactions link the molecules into a C(7) chain along the [20\(\bar {1}\)] direction. The crystal packing is also stabilized by N–O⋅⋅⋅π interaction. The molecular structure and vibrational frequencies of the title compound were calculated using the DFT/B3LYP method with the 6-311++G(d,p) basis set. The calculated results show that the optimized geometry can well reproduce the crystal structure, and the theoretical vibrational frequencies show good agreement with experimental results.





Similar content being viewed by others
REFERENCES
H. Schiff, Justus Liebigs Ann. Chem. 131, 118 (1864).
D. Barton and W. D. Ollis, Comprehensive Organic Chemistry (Pergamon, Oxford, 1979).
R. W. Layer, Chem. Rev. 63, 489 (1963).
C. K. Ingold, Structure and Mechanism in Organic Chemistry (Cornell Univ., Ithaca, 1969).
H. Tanak, A. A. Agar, and M. Yavuz, J. Mol. Model. 16, 577 (2010).
A. A. Jarrahpour, A. F. Jalbout, S. Rezaei, et al., Molbank 2006, M455 (2006).
A. A. Jarrahpour, M. Shekarriz, and A. Taslimi, Molecules 9, 29 (2004).
A. E. Taggi, A. M. Hafez, H. Wack, et al., J. Am. Chem. Soc. 124, 6626 (2002).
K. I. Molvi, K. K. Vasu, S. G. Yerande, et al., Eur. J. Med. Chem. 42, 1049 (2007).
N. Satheesha Rai, B. Kalluraya, B. Lingappa, et al., Eur. J. Med. Chem. 43, 1715 (2008).
B. V. Asthalatha, B. Narayana, K. K. Vijaya Raj, et al., Eur. J. Med. Chem. 42, 719 (2007).
B. Kalluraya, A. D’Souza, and B. S. Holla, Indian J. Chem. Sect. B 33, 1017 (1994).
B. Kalluraya and S. N. Shetty, Indian J. Heterocycl. Chem. 6, 287 (1997).
H. K. Fun, S. Arshad, S. Shetty, et al., Acta Crystallogr. E 68, o2681 (2012).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., GAUSSIAN 09, Revision C.01 (Gaussian, Wallingford, CT, 2009).
R. Dennington II, T. Keith, and J. Millam, Gauss View, Version 5.0 (Semichem Inc., Shawnee Mission, KS, 2008).
B. A. Bruker, Inc., Madison, Wisconsin, USA, 2007.
G. M. Sheldrick, Acta Crystallogr. A 64, 112 (2007).
L. J. Farrugia, J. Appl. Crystallogr. 30, 565 (1997).
H. Tanak, F. Koçak, and E. Agar, Mol. Phys. 114, 197 (2016).
F. Koçak, H. Tanak, E. Ağar, et al., Acta Crystallogr. E 71, o418 (2015.
Ş. Karataş, H. Tanak, and A. A. Ağar, Crystallogr. Rep. 61, 410 (2016).
U. Ceylan, H. Tanak, S. Gumus, et al., Acta Crystallogr. E 67, o2004 (2011).
U. Ceylan, S. Gumus, E. Ağar, et al., Acta Crystallogr. E 68, o2116 (2012).
J. Bernstein, R. E. Davies, L. Shimoni, et al., Angew. Chem. Int. Ed. Engl. 34, 1555 (1995).
U. Ceylan, G. O. Tarı, H. Gökçe, and E. Ağar, J. Mol. Struct. 1110, 1 (2016).
İ. Bozkurt, M. Evecen, H. Tanak, et al., J. Mol. Struct. 1197, 9 (2019).
Funding
This study was supported financially by the Research Centre of Ondokuz Mayıs University (project no. PYO.FEN.1904.18.003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasan Tanak, Karataş, Ş., Meral, S. et al. Crystal Structure, Spectroscopic and DFT Computational Studies of N-(4-Fluorophenyl)-1-(5-Nitrothiophen-2-yl)methanimine. Crystallogr. Rep. 65, 1221–1225 (2020). https://doi.org/10.1134/S1063774520070263
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774520070263