Abstract
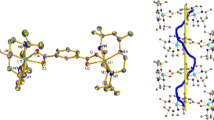
The structural aspects of a family of S-alkylated complexes, generated by reacting iodoacetamide or iodoethanol with two mononuclear Ni(II) diaminodithiolate complexes are discussed. The S-alkylation reactions were investigated with particular attention paid to the size of the chelate ring that straps the N,N′-methylamine donors. In one complex the N-methyl groups are cis to each other and in the other they are trans. Both complexes undergo S-alkylation with two equivalents of either reagent, that coordinates through the pendant oxygen to the Ni(II), forming dications with an N2S2O2 ligand donor set. Crystal structures of [NiC12H26N4O2S2]I2 · MeOH, [NiC12H28N2O2S2]I2, and [NiC13H30N2O2S2]I2 · 1/2MeOH, are determined by single crystal X-ray analysis. The N-methyl groups in each of the alkylated derivatives are trans to each other, suggesting that the cis configuration is highly unfavored for such complexes in octahedral conformation. Crystal packing data shows that each of the alkylated complexes interacts closely with the iodide counterions, and with solvent if present; some of these interactions include H-bonds. Only the iodoacetamide derivative shows any significant interaction with a neighboring molecule.
Similar content being viewed by others
References
Z. Hu, N. J. Spangler, M. E. Anderson, J. Xia, P. W. Ludden, P. A. Lindahl, and E. Münck, J. Am. Chem. Soc. 118, 830 (1996).
V. J. DeRose, J. Telser, M. E. Anderson, P. A. Lindahl, and B. M. Hoffman, J. Am. Chem. Soc. 120, 8767 (1998).
G. O. Tan, S. A. Ensign, S. Ciurli, M. J. Scott, B. Hedman, R. H. Holm, P. W. Ludden, Z. R. Korszun, P. J. Stephens, and K. O. Hodgson, Proc. Nat. Acad. Sci. USA. 89, 4427 (1992).
J. Xia, J. Dong, S. Wang, R. A. Scott, and P. A. Lindahl, J. Am. Chem. Soc. 117, 7065 (1995).
C. Y. Ralston, H. Wang, S. W. Ragsdale, M. Kumar, N. J. Spangler, P. W. Ludden, W. Gu, R. M. Jones, D. S. Patil, and S. P. Cramer, J. Am. Chem. Soc. 122, 10553 (2000).
S. W. Ha, M. Korbas, M. Klepsch, W. Meyer-Klaucke, O. Meyer, and V. Svetlitchnyi, J. Biol. Chem. 282, 10639 (2007).
S. W. Ragsdale and E. Pierce, Biochim. Biophys. Acta 1784, 1873 (2008).
S. B. Choudhury, J. W. Lee, G. Davidson, Y. I. Yim, K. Bose, M. L. Sharma, S. O. Kang, D. E. Cabelli, Biochemistry 38, 3744 (1999).
H. D. Youn, E. J. Kim, J. H. Roe, Y. C. Hah, and S. O. Kang, Biochem. J. 318, 889 (1996).
D. P. Barondeau, C. J. Kassmann, C. K. Bruns, J. A. Tainer, and E. D. Getzoff, Biochemistry 43, 8038 (2004).
A. Volbeda, M-H. Charon, C. Piras, E. C. Hatchikian, M. Frey, and J. C. Fontecilla-Camps, Nature 373, 580 (1995).
A. Volbeda, E. Garcin, C. Piras, A. L. deLacey, V. M. Fernandez, E. C. Hatchikian, M. C. Frey, and J. C. Fontecilla-Camps, J. Am. Chem. Soc. 118, 12989 (1996).
J. C. Fontecilla-Camps, A. Volbeda, C. Cavazza, and Y. Nicolet, Chem. Rev. 107, 4273 (2007).
P. M. Vignais and B. Billoud, Chem. Rev. 107, 4206 (2007).
G. M. Sheldrick, Acta Crystallogr. A 46, 467 (1990).
L. J. Farrugia, J. Appl. Crystallogr. 30, 565 (1997).
Mercury CSD 3. 1. 1 (Build RC7) (Cambridge Crystallographic Data Centre, Cambridge, 2013).
A. J. Gordon and R. A. Ford, The Chemist’s Companion (Wiley, New York, 1972).
J. A. Riddick, W. B. Bunger, and T. K. Sakano, Organic Solvents, 4th ed. (Wiley, New York, 1986).
W. J. Hu, D. Barton, and S. J. Lippard, J. Am. Chem. Soc. 95, 1170 (1973).
J. L. Corbin, K. F. Miller, N. Pariyadath, S. Wherland, A. E. Bruce, and E. I. Stiefel, Inorg. Chim. Acta, 90, 41 (1984).
M. A. Turner, W. L. Driessen, and J. Reedijk, Inorg. Chem. 29, 3331 (1990).
E. R. Hosler, R. W. Herbst, B. S. Chohan, and M. J. Maroney, RSC Dalton Trans. 41, 804 (2012).
S. J. Lippard, Acc. Chem. Res. 6, 282 (1973).
K. D. Karlin and S. J. Lippard, J. Am. Chem. Soc. 98, 6951 (1976).
G. J. Colpas, M. Kumar, R. O. Day, and M. J. Maroney, Inorg Chem. 29, 4779 (1990).
H. F. Crouse, J. Potoma, F. Nejrabi, D. L. Snyder, B. S. Chohan, and S. Basu, RSC Dalton Trans. 41, 2720 (2012).
D. Gibson and S. J. Lippard, Inorg. Chem. 25, 219 (1986).
A. Avdeef, F. Hartenstein, A. R. Chemotti, and J. A. Brown, Inorg. Chem. 31, 3701 (1992).
J. Yoo, J. Ko, and S-E. Park, Bull. Korean. Chem. Soc. 15, 803 (1994).
J. J. Smee, D. C. Goodman, J. H. Reibenspies, and M. Y. Darensbourg, Eur. J. Inorg. Chem. 539 (1999).
B. S. Chohan, Crystallogr. Rep. 58, 1067 (2013).
D. C. Goodman, R. M. Buonomo, P. J. Farmer, J. H. Reibenspies, and M. Y. Darensbourg, Inorg. Chem. 35, 4029, (1996).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Chohan, B.S. Structural features of a series of S-alkylated and non-S-alkylated aminothiolate nickel(II) complexes. Crystallogr. Rep. 59, 979–986 (2014). https://doi.org/10.1134/S1063774514070086
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774514070086