Abstract
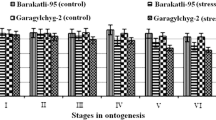
Changes in the enzymatic activity of phenol-dependent peroxidase (PO) of vacuoles and tissue extract of red beet (Beta vulgaris L.) roots in different phases of plant development and in hyperosmotic stress and pathogen infection were found. The highest activity was observed during root growth and the lowest PO activity occurred in dormancy, respectively. Activation of the enzyme was observed in infected roots. The isozyme composition of PO was characterized by lability, and the number of cationic isoforms varied significantly. The optimum pH of the enzyme changed depending on the growth phase and stressor, tending to shift towards low values at rest and in hyperosmotic stress. The shift in the optimum pH coincided with the appearance of additional cationic PO isoforms.
Similar content being viewed by others
References
Andreeva, V.A., Ferment peroksidaza: uchastie v zashchitnom mekhanizme rastenii (Peroxidase: Involvement in Plant Defense Mechanism), Moscow: Nauka, 1988.
Andrews, J., Adams, S.R., and Burton, K.S., Evered C.E. Subcellular localization of peroxidase in tomato fruit skin and the possible implications for the regulation of fruit growth, J. Exp. Bot., 2002, vol. 53, pp. 2185–2191.
Bernal, M.A., Pedreno, M.A., Calderón, A.A., et al., Purification of a basic peroxidase isoenzyme from capsicum fruits and the immunoinhibition of its capsaicin oxidation capacity by antibodies raised against horseradish peroxidase, Z. Lebensm. Unters Forsch., 1994, vol. 199, pp. 240–242.
Bradford, M., A rapid and sensitive method for the quantitation of protein utilising the principal of protein-dye binding, Anal. Biochem., 1976, vol. 72, pp. 248–254.
Cruz de Carvalho, M., Drought stress and reactive oxygen species, Plant Signal Behav., 2008, vol. 3, pp. 156–165.
Costa, M.M.R., Hilliou, F., Duarte, P., et al., Molecular cloning and characterization of a vacuolar class III peroxidase involved in the metabolism of anticancer alkaloids in Catharanthus roseus, Plant Physiol., 2008, vol. 146, pp. 403–417.
Crèvecoeur, M., Pinedo, M., Greppin, H., and Penel, C., Peroxidase activity in shoot apical meristem from Spinacia, Acta Histochem., 1997, vol. 99, pp. 177–186.
Dolgova, L.G., Peroxidase activity—an indicator of resistance of introduced plants in the steppe zone of Ukraine, Vestn. Dnepropetrovsk. Nats. Univ., 2004, vol. 12, no. 1, pp. 38–41.
Dowd, P.F. and Johnson, E.T., Association of a specific cationic peroxidase isozyme with maize stress and disease resistance responses, genetic identification, and identification of a cDNA coding for the isozyme, J. Agric Food Chem., 2005, vol. 53, pp. 4464–4470.
Escribano, J., Gandia-Herrero, F., Caballero, N., and Pedreno, M.A., Subcellular localization and isoenzyme pattern of peroxidase and polyphenol oxidase in beet root (Beta vulgaris L.), J. Agric Food Chem., 2002, vol. 50, pp. 6123–6129.
Fu, J. and Huang, B., Involvement of antioxidants and lipid peroxidation in the adaptation of two cool-season grasses to localized drought stress, Environ. Exp. Bot., 2001, vol. 45, pp. 105–114.
Isheeva, O.D., Pradedova, E.V., and Salyaev, R.K., Multiplicity of vacuolar peroxidase isozymes in higher plants by the example of red beet (Beta vulgaris L.) roots, Doklady Biochem. Biophys., 2009, vol. 424, pp. 53–55.
Kim, S.S. and Lee, D.J., Purification and characterization of a cationic peroxidase Cs in Raphanus sativus, Plant Physiol., 2005, vol. 162, pp. 609–617.
Kolupaev, Yu.E., Reactive oxygen species in plants under the action of stressors: formation and possible functions, Vestn. Khar’k. Nats. Agrarn. Univ., Ser. Biol., 2007, no. 12, pp. 6–26.
Kristensen, B.K., Bloch, H., and Rasmussen, S.K., Barley coleoptile peroxidases. purification, molecular cloning, and induction by pathogens, Plant Physiol., 1999, vol. 120, pp. 501–512.
Kuzevanov, V.Ya., Katkov, B.B., and Salyaev, R.K., General principles of isolation of vacuoles and vacuolar membranes, in Struktura i funktsii biologicheskikh membran rastenii (Structure and Functions of Biological Membranes of Plants), Salyaev, R.K. and Voinikov, V.K., Eds., Novosibirsk: Nauka, 1985, pp. 93–107.
López-Molina, D., Heering, H.A., Smulevich, G., et al., Purification and characterization of a new cationic peroxidase from fresh flowers of Cynara scolymus L., J. Inorg. Biochem., 2003, vol. 94, pp. 243–254.
López-Serrano, M., Ferrer, M.A., Barceló, R.A., and Pedreno, M.A., Effect of fosetyl-A1 on peroxidase from grapevine (Vitis vinifera) cells, Eur. J. Histochem., 1995, vol. 39, pp. 69–74.
Martinez-Parra, J. and Munoz, R., Characterization of betacyanin oxidation catalyzed by a peroxidase from Beta vulgaris L. roots, J. Agric. Food Chem., 2001, vol. 49, pp. 4064–4068.
Marty, F., Plant vacuoles, Plant Cell, 1999, vol. 11, pp. 587–599.
Minibaeva, F.V. and Gordon, L.Kh., Superoxide production and the activity of extracellular peroxidase in plant tissues under stress conditions, Russ. J. Plant Physiol., 2003, vol. 50, no. 3, pp. 411–417.
Mohamed, S.A., El-Badry, M.O., Drees, E.A., and Fahmy, A.S., Properties of a cationic peroxidase from Citrus jambhiri cv. Adalia, Appl. Biochem. Biotechnol., 2008, vol. 150, pp. 127–137.
Naumov, N.A., Bolezni sel’skokhozyaistvennykh rastenii (fitopatologiya) (Diseases of Agricultural Plants (Plant Pathology)), Moscow: Sel’khozgiz, 1940.
Osterman, L.A., Issledovanie biologicheskikh makromolekul elektrofokusirovaniem, immunoelektroforezom i radioizotopnymi metodami (Diseases of Agricultural Plants (Plant Pathology)), Moscow: Nauka, 1983.
Pobezhimova, T.P., Kolesnichenko, A.V., and Grabel’nykh, O.I., Metody izucheniya mitokhondrii rastenii. Polyarografiya i elektroforez (Methods of Studying Plant Mitochondria: Polarography and Electrophoresis), Moscow: NPK Promekobezopasnost’, 2004.
Polesskaya, O.G., Rastitel’naya kletka i aktivnye formy kisloroda (Plant Cell and Reactive Oxygen Species), Moscow: KDU, 2007.
Pradedova, E.V., Isheeva, O.D., and Salyaev, R.K., Superoxide dismutase of plant cell vacuoles, Biol. Membr., 2009, vol. 26, no. 1, pp. 21–30.
Ranieri, A., Castagna, A., Baldan, B., and Soldatini, G.F., Iron deficiency differently affects peroxidase isoforms in sunflower, J. Exp. Bot., 2001, vol. 52, pp. 25–35.
Salyaev, R.K., Kuzevanov, V.Ya., Khaptagae, C.B., and Kopytchuk, V.N., Isolation and purification of vacuoles and vacuolar membranes of plant cells, Fiziol. Rast., 1981, vol. 28, pp. 1295–1305.
Sottomayor, M., Barceló, A.R., and Sottomayor, M., Peroxidase from Catharanthus roseus (L.) G. Don and the biosynthesis of alpha-3′,4′-anhydrovinblastine: a specific role for a multifunctional enzyme, Protoplasma, 2003, vol. 222, pp. 97–105.
Thakur, P.S., Singh, G., and Rai, V.K., Peroxidase isozymes in relation to developing water deficits in two Zea mays L. cultivars, New Phytol., 1981, vol. 89, pp. 25–32.
Van den Berg, B.M., Chibbar, R.N., and van Huystee, R.B., A comparative study of a cationic peroxidase from peanut and an anionic peroxidase from petunia, Plant. Cell. Rep., 1983, vol. 2, pp. 304–307.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.V. Pradedova, O.D. Nimaeva, R.K. Salyaev, 2014, published in Izvestiya Akademii Nauk, Seriya Biologicheskaya, 2014, No. 3, pp. 254–263.
Rights and permissions
About this article
Cite this article
Pradedova, E.V., Nimaeva, O.D. & Salyaev, R.K. Effect of stress conditions on the activity and isozyme composition of peroxidase of vacuoles and tissue extract of red beet roots. Biol Bull Russ Acad Sci 41, 233–241 (2014). https://doi.org/10.1134/S106235901403008X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106235901403008X