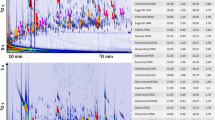
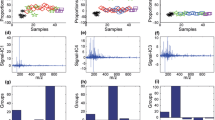
Abstract
In this study, the idea of sample-specific metabolites library with retention neighbor was presented for better identification and quantitation of metabolites. Compared with universal libraries, the established library was found to have significantly improved match indices (similarity, reverse, and probability), indicating better identification of metabolites. For quantitative metabolic profiling of temperature treated tobacco samples, the established method integrated and aligned 175 metabolites with no missing value, better than the widely used metabolomics software including XCMS Online, MetaboliteDetector, SpectConnect, and ChromaTOF. Partial least squares discriminant analysis models were also used for quantitative evaluation and the established method produced better values of model parameters than the compared software, indicating a better model was established using the present method.





Similar content being viewed by others
REFERENCES
Fiehn, O., Plant Mol. Biol., 2002, vol. 48, nos. 1–2, p. 155.
Kind, T., Wohlgemuth, G., Lee, D.Y., Lu, Y., Palazoglu, M., Shahbaz, S., and Fiehn, O., Anal. Chem., 2009, vol. 81, no. 24, p. 10038.
Smith, C.A., Want, E.J., O’Maille, G., Abagyan, R., and Siuzdak, G., Anal. Chem., 2006, vol. 78, no. 3, p. 779.
Hiller, K., Hangebrauk, J., Jager, C., Spura, J., Schreiber, K., and Schomburg, D., Anal. Chem., 2009, vol. 81, no. 9, p. 3429.
Tsugawa, H., Cajka, T., Kind, T., Ma, Y., Higgins, B., Ikeda, K., Kanazawa, M., van der Gheynst, J., Fiehn, O., and Arita, M., Nat. Methods, 2015, vol. 12, no. 6, p. 523.
Tikunov, Y.M., Laptenok, S., Hall, R.D., Bovy, A., and Vos, R.C.H., Metabolomics, 2012, vol. 8, no. 4, p. 714.
Styczynski, M.P., Moxley, J.F., Tong, L.V., Walther, J.L., Jensen, K.L., and Stephanopoulos, G.N., Anal. Chem., 2007, vol. 79, no. 3, p. 966.
Warren, C.R., Metabolomics, 2013, vol. 9, no. 1, p. 110.
Kartsova, L.A. and Solov’eva, S.A., J. Anal. Chem., 2019, vol. 74, no. 4, p. 307.
Wiley Registry™ of Mass Spectral Data, 12th ed. http://www.sisweb.com/software/wiley-registry.htm. Accessed October 8, 2019.
Lin, S., Liu, N., Yang, Z., Song, W., Wang, P., Chen, H., Lucio, M., Schmitt-Kopplin, P., Chen, G., and Cai, Z., Talanta, 2010, vol. 83, no. 1, p. 262.
Kopka, J., Schauer, N., Krueger, S., Birkemeyer, C., Usadel, B., Bergmüller, E., Dormann, P., Weckwerth, W., Gibon, Y., Stitt, M., Willmitzer, L., Fernie, A.R., and Steinhauser, D., Bioinformatics, 2005, vol. 21, no. 8, p. 1635.
Gika, H.G., Macpherson, E., Theodoridis, G.A., and Wilson, I.D., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2008, vol. 871, no. 2, p. 299.
Gika, H.G., Theodoridis, G.A., Wingate, J.E., and Wilson, I.D., J. Proteome Res., 2007, vol. 6, no. 8, p. 3291.
Sangster, T., Major, H., Plumb, R., Wilson, A.J., and Wilson, I.D., Analyst, 2006, vol. 131, no. 10, p. 1075.
Li, Y., Pang, T., Li, Y., Wang, X., Li, Q., Lu, X., and Xu, G., J. Sep. Sci., 2011, vol. 34, no. 12, p. 1447.
Li, Y., Ruan, Q., Ye, G., Lu, X., Lin, X., and Xu, G., J. Chromatogr. A, 2012, vol. 1255, p. 228.
Lisec, J., Schauer, N., Kopka, J., Willmitzer, L., and Fernie, A.R., Nat. Protoc., 2006, vol. 1, no. 1, p. 387.
Lee, D.Y. and Fiehn, O., Plant Methods, 2008, vol. 406, no. 28, p. 7275.
Fiehn, O., TrAC, Trends Anal. Chem., 2008, vol. 27, no. 3, p. 261.
Szopa, J., Wilczyński, G., Fiehn, O., Wenczel, A., and Willmitzer, L., Phytochemistry, 2001, vol. 58, no. 2, p. 315.
Fiehn, O., Phytochemistry, 2003, vol. 62, no. 6, p. 875.
Li, Y., Pang, T., Li, Y., Ye, G., Lu, X., and Xu, G., J. Sep. Sci., 2013, vol. 36, nos. 9–10, p. 1545.
Golm Metabolome Database. http://gmd.mpimp-golm.mpg.de. Accessed October 8, 2019.
Mass Spectrometry Data Center. http://chemdata.nist. gov/mass-spc/ms-search/docs/Ver20Man_11.pdf. Accessed October 8, 2019.
Vandendool, H. and Kratz, P.D., J. Chromatogr., 1963, vol. 11, no. 4, p. 463.
XCMS, The original and most widely used metabolomic platform. https://xcmsonline.scripps.edu/landing_page.php?pgcontent=mainPage. Accessed October 8, 2019.
Benton, H.P., Wong, D.M., Trauger, S.A., and Siuzdak, G., Anal. Chem., 2008, vol. 80, no. 16, p. 6382.
Gowda, H., Ivanisevic, J., Johnson, C.H., Kurczy, M.E., Benton, H.P., Rinehart, D., Nguyen, T., Ray, J., Kuehl, J., Arevalo, B., Westenskow, P.D., Wang, J., and Arkin, A.P., Anal. Chem., 2014, vol. 86, no. 14, p. 6931.
Wang, B., Fang, A., Heim, J., Bogdanov, B., Pugh, S., Libardoni, M., and Zhang, X., Anal. Chem., 2010, vol. 82, no. 12, p. 5069.
Koh, Y., Pasikanti, K.K., Yap, C.W., and Chan, E.C., J. Chromatogr. A, 2010, vol. 1217, no. 52, p. 8308.
Lu, H., Dunn, W.B., Shen, H., Kell, D.B., and Liang, Y., TrAC, Trends Anal. Chem., 2008, vol. 27, no. 3, p. 215.
Gromski, P.S., Muhamadali, H., Ellis, D.I., Xu, Y., Correa, E., Turner, M.L., and Goodacre, R., Anal. Chim. Acta, 2015, vol. 879, p. 10.
Multi- and Megavariate Data Analysis. Part I: Basic Principles and Applications, Eriksson, L., Johansson, E., Kettaneh-Wold, N., Trygg, J., Wikstrom, C., and Wold, S., Eds., Stockholm: UMETRICS AB, 2006.
ACKNOWLEDGMENTS
The authors thank the staff of the chemistry research center of Yunnan academy of tobacco agricultural sciences for their important contributions.
Funding
The study was supported by the foundation (nos. 20185300002410027 and 2018530000241007) from China National Tobacco Company Yunnan branch.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Yong Li, Pang, T., Shi, JL. et al. Sample-Specific Metabolites Library with Retention Neighbor: an Improved Identification and Quantitation Strategy for Gas Chromatography–Mass Spectrometry-Based Metabolomics. J Anal Chem 76, 844–853 (2021). https://doi.org/10.1134/S1061934821070108
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934821070108