Abstract
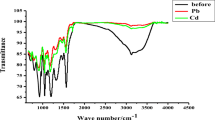
The construction and performance characteristics of novel PVC membrane (PME) and coated platinum (CPtE) cadmium ion selective electrodes based on N,N′-bis(salicylaldehyde)phenylenediamine (salophen) are described. The electrodes exhibit linear responses along with near Nernstian slopes of 28.9 ± 0.4 (PME) and 29.2 ± 0.6 (CPtE) mV/decade of concentration within the Cd2+ ion concentration range of 5.7 × 10−8 to 3.7 × 10−3 M for PME and 3.0 × 10−8–3.0 × 10−3 M for CPtE. These sensors are applicable in a pH range of 2.5–7.5. The lower detection limits by PME and CPtE are 3.2 × 10−8 and 1.6 × 10−8 M, respectively. They have a response time less than 14 s and can be used practically for a period of at least 2 months without any measurable divergence in results. The electrode can also tolerate partially non-aqueous media (ethanol, methanol and acetone) up to 30%. The electrodes showed excellent selectivity towards Cd2+ ion over a wide range of alkali, alkaline earth, and transition metals ions. They were successfully applied to the direct determination of Cd2+ ions in tap water, aqueduct water, and river water and soil sample. In addition the electrodes were used as an indicator electrode in potentiometric titration of Cd2+ ion with EDTA.
Similar content being viewed by others
References
Goering, P.L., Waalkes, M.P. and Klaassen, C.D. /Handbook of Experimental Pharmacology: Toxicology of Metals, Biochem. Effects / Eds. Goyer R.A., Cherian M.G., vol. 115, New York: Springer-Verlag, 1994, p. 189.
Satarug, S., Baker, J.R., Urbenjapol, S., Haswell-Elkins, M., Reilly, D.J., Williams, P.E.B., and Moore, M.R., Toxicol. Lett. 2003, vol. 137, p. 65.
Waalkes, M.P., J. Inorg. Biochem., 2000, vol. 79, p. 241.
Tenth report on Carcinogens, Department of Health, and Human Services, National Toxicology Program, Research triangle park, NC, 42, 2000.
Wen, X., Wu, P., Chen, L., and Hou, X., Anal. Chim. Acta, 2009, vol. 650, p. 33.
Vilar Farinas, M., Barciela Garcıa, J., García Martín, S., Pena Crecente, R., and Herrero Latorre, C., Anal. Chim. Acta, 2007, vol. 591, p. 231.
Potts, L.W., Quantitative Analysis. Theory and Practice, New York: Harper and Row, 1987.
Davis, A.C., Calloway Jr., C.P., and Jones, B.T., Talanta, 2007, vol. 71, p. 1144.
Yi, W. J., Li, Y., Ran, G., Luo, H.Q., and Li, N.B., Sens. Actuat. B, 2012, vol. 166–167, p. 544.
Este-vez-Herna-ndez, O., Naranjo-Rodríguez, I., Hidalgo-Hidalgo de Cisneros, J.L., and Reguera, E., Sens. Actuat. B, 2007, vol. 123, p. 488.
Hong-Xia, Y., Wei, L., Bing, L., Wei, W., Hui-Juan, Z., and Deng-Yun, C., Chin. J. Anal. Chem., 2009, vol. 37, p. 1511.
Marshall, G.D. and Van Staden, J.F., Instrum. Sci. Technol., 1997, vol. 25, p. 307.
Gupta, V.K., Singh, A.K., and Gupta, B., Anal. Chim. Acta, 2007, vol. 340, p. 583.
Tyagi, S., Agarwal, H., and Ikram, S., Water Sci. Technol. 2010, vol. 62, p. 2510.
Rosty, R., Kebbekus, B., and Zaitsev, V., Sens. Actuators B, 2005, vol. 107, p. 347.
Ryabushko, O.P., Pilipenko, A.T., Savin, Yu.S., and Batkovskaya, L.A., Ukr. Khim. Zh., 1990, vol. 56, p. 263.
Javanbakht, M., Shabanikia, A., Darvich, M.R., Ganjali, M.R., and Shamsipur, M., Anal. Chim. Acta, 2000, vol. 408, p. 75.
Khamjumphol, U., Watchasit, S., Suksai, C., Janrungroatsakul, W., Boonchiangma, S., and Tuntulani, T., Anal. Chim. Acta, 2011, vol. 704, p. 73.
Gupta, V.K., and Kumar, P., Anal. Chim. Acta, 1999, vol. 389, p. 205.
Gupta, V.K., Jain, A.K., and Kumar, P., Electrochim. Acta, 2006, vol. 52, p. 736.
Pineros, M.A., Shaff, J.E., Kochian, L.V., and Bakker, E., Electroanal., 1998, vol. 10, p. 937.
Amirnasra, M., Schenkb, K.J., Gorjic, A., and Vafazadehd, R., Polyhedron, 2001, vol. 20, p. 695.
Bakker, E., Buhlmann, P., and Pretsch, E., Chem. Rev., 1997, vol. 97, p. 3083.
Shamsipur, M., Rouhani, S., Sharghi, H., Ganjali, M.R., and Eshghi, H., Anal. Chem., 1999, vol. 71, p. 4983.
Gupta, K.C. and D’Arc, M.J., Anal. Chim. Acta, 2001, vol. 437, p. 199.
Gupta, V.K., Goyal, R.N., Jain, A.K., and Sharma, R.A., Electrochim. Acta, 2009, vol. 54, p. 3218.
IUPAC Analytical Chemistry Division. Commission on Analytical Nomenclature. Recommendations for Namenclature of Ion-Selective Electrodes, Pure Appl. Chem. 1976, vol. 48, p. 127.
Shamsipur, M., Hosseini, M., Alizadeh, K., Talebpour, Z., Mousavi, M.F., Ganjali, M.R., Arca, M., and Lippolis, V., Anal. Chem., 2003, vol. 75, p. 5680.
Bakker, E., Pretsch, E., and Buhlman, P., Anal. Chem., 2000, vol. 72, p. 1127.
Bakker, E., Anal. Chem., 1997, vol. 69, p. 1061.
Gadzekpo, V.P.Y. and Christian, G.D., Anal. Chim. Acta., 1984, vol. 164, p. 279.
Umezawa, Y., Umezawa, K., and Sato, H., Pure Appl. Chem. 1995, vol. 67, p. 507.
Ghanei-Motlagh, M., Taher, M.A., Saheb, V., Fayazi, M., and Sheikhshoaie, I., Electrochim. Acta, 2011, vol. 56, p. 5376.
Gupta, V.K., Singh, A.K., and Gupta, B., Anal. Chim. Acta, 2007, vol. 583, p. 340.
Gupta, V.K., Jain, A.K., Ludwig, R., and Maheshwari, G., Electrochim. Acta, 2008, vol. 53, p. 2362.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Mirzaei, M., Pili, H.B. Potentiometric determination of cadmium using coated platinum and PVC membrane sensors based on N,N′-bis(salicylaldehyde)phenylenediamine (salophen). J Anal Chem 70, 731–737 (2015). https://doi.org/10.1134/S1061934815060076
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934815060076