Abstract
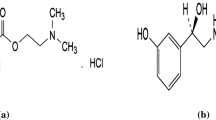
Betamethasone sodium phosphate (BMNaP) has been employed as an electroactive material in the design of an ion-selective electrode (ISE). The electrode incorporates PVC membrane with betamethasone sodium phosphate-lidocaine ion pair complex. The influences of membrane composition, temperature, pH of the test solution, and the interfering ions on the electrode performance were investigated. The sensor exhibits a Nernstian response for betamethasone sodium phosphate ions over a relatively wide concentration range (1.0 × 10−1 to 1.0 × 10−5 M) with a slope of 28.4 ± 0.9 mV per decade at 25°C. It can be used in the pH range 4.0–10.0. The isothermal temperature coefficient of this electrode amounted to −0.0008 V/°C. The membrane sensor was successfully applied to the determination of betamethasone sodium phosphate in pharmaceutical products.
Similar content being viewed by others
References
Bühlmann, P., Pretsch, E., and Bakker, E., Chem. Rev., 1998, vol. 98, p. 1593.
Mahajan, R.K., Kumar, M., Sharma, V., and Kaur, I., Analyst, 2001, vol. 126, p. 505.
Srivastva, S.K., Gupta, V.K., and Jain, S., Anal. Chem., 1996, vol. 68, p. 1272.
Diamond, D., J. Incl. Phenom. Macrocycl. Chem., 1994, vol. 19, p. 149.
Giahi, M., Aghaie, H., Arvand, M., and Hejri, A.M., Russian Journal of Electrochemistry, 2005, vol. 41, p. 1290.
Aghaie, H., Giahi, M., Monajjemi, M., Arvand, M., Nafissi, G.H., and Aghaie, M., Sens Actuators B, 2005, vol. 107, p. 756.
Arvand, M., Pourhabib, A. Shemshadi, and Giahi, M., Anal. Bioanal. Chem., 2007, vol. 387, p. 1033.
Aghaie, M., Giahi, M., Aghaie, H., and Etryan, A., Russian Journal of Electrochemistry 2009, vol. 45, p. 804.
Aghaie, M., Giahi, M., Aghaie, H., Arvand, M., Pournaghdy, M., and Yavari, F., Desalination, 2009, vol. 251, p. 346.
Cosofret, V.V., Membrane Electrodes in Drug Substances Analysis, Pergamon Press, Oxford, 1982.
Campanella, L. and Tomasseti, M., Sel. Electrode Rev., 1989, vol. 11, p. 69.
Cosofret, V.V. and Buck, R.P., Crit. Rev. Anal. Chem., 1993, vol. 24, p. 1.
Valsami, G.N., Macheras, P.E., and Koupparis, M.A., Analyst, 1989, vol. 114, p. 387.
Valsami, G.N., Koupparis, M.A., and Macheras, P.E., Pharmaceut. Res., 1992, vol. 9, p. 94.
Hassan, S.S., Amer, M.M., Abd. El-Fatah, S.A., and El-Kosasy, A.M., Anal. Chim. Acta, 1998, vol. 363, p. 81.
Giahi, M., Arvand, M., Mirzaei, M., Bagherinia, M.A., Anal. Lett., 2009, vol. 42, p. 870.
Giahi, M., Pournaghdy, M., and Rakhshaee, R., J. Anal. Chem., 2009, vol. 64, p. 188.
Giahi, M. Mirzaei M. and Veghar, G., J. Iranian Chem. Soc., 2010, vol. 7, p. 333.
Kharitonov, S.V., J. Pharm. Biom. Anal., 2002, vol. 30, p. 181.
Moody, G.J., Oke, R.B., and Thomas, J.D.R., Analyst, 1970, vol. 95, p. 910.
Gutknecht, J., Schneider, H., and Stroka, J., Inorg. Chem., 1978, vol. 17, p. 3326.
Matysik, S., Matysik, F.M., Mattusch, J., and Einicke, W.D., Electroanalysis, 1998, vol. 10, p. 98.
Bakker, E., Bühlmann, P., and Pretsch, E., Chem. Rev., 1997, vol. 97, p. 3083.
Gadzepko, V.P. and Christian, G.D., Anal. Chim. Acta, 1984, vol. 164, p. 279.
Antropov, L.I., Theoretical Electrochemistry, Mir, Moscow, 1972.
Mostafa, G.A.E., Anal. Sci., 2002, vol. 18, p. 1335.
Jalali, F., Rajabi, M., Bahrami, G., and Shamsipur, M., Anal. Sci., 2005, vol. 21, p. 1533.
Wang, L., Yang, Y.Y., Chung, T.S., and Chen, X.Q., J. Pharm. Biom. Anal., 2002, vol. 28, p. 629.
Singh, D.K. and Verma, R. Iranian J. Pharmacology & Therapeutics, 2008, vol. 7(1), p. 61.
Author information
Authors and Affiliations
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Giahi, M., Marvi, O., Safari, F. et al. Determination of betamethasone sodium phosphate in pharmaceuticals by potentiometric membrane sensor based on its lidocaine ion pair. J Anal Chem 68, 900–905 (2013). https://doi.org/10.1134/S1061934813100067
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934813100067