Abstract
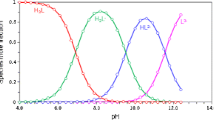
The comparison of literature data on the dissociation constant of one of the most abundant natural flavonoids, e.g., quercetin, demonstrates their irreproducibility. The reason for this likely corresponds to the easiness of its oxidation by air oxygen during the titration process. To eliminate such problems, a modified version of potentiometric titration was proposed with bubbling a weak flow of an inert gas (nitrogen) through the solution to be titrated in the presence of minimal amounts of a nonionic surfactant. By virtue of the technique proposed, the values of pK a for quercetin were measured to be 6.62 ± 0.04 and 9.7 ± 0.3. The first one corresponds to the hydroxyl group in the γ-pyrone fragment of the molecule, while another agrees with the typical values of pK a for phenols.
Similar content being viewed by others
References
Flavonoids: Chemistry, Biochemistry and Applications, Andersen, O.M. and Markham, K.R., Eds., New York: Taylor & Francis, 2005.
The Science of Flavonoids, Grotewold, E., Ed., New York: Springer, 2006.
Khimicheskaya entsiklopediya (Chemical Encyclopedia), Moscow: Bol’shaya Sovetskaya Encyklopediya, 1998, vol. 5.
Rastitel’nye resursy SSSR, (Plant Resources of the USSR), vols. 1–8, Leningrad: Nauka, 1984–1994.
Simpson, T.N. and Beton, J.L., J. Chem. Soc., 1954, p. 4065.
Tekhnologiya i standartizatsiya lekarstv (Technology and Standardization of Drugs), Georgievskii, V.P. and Konev, F.A., Eds., Kharkov: RIREG, 1996.
Tyukavkina, N.A. and Pogodaeva, N.N., Khim. prirodn. soed., 1971, no. 1, p. 11.
Georgievskii, V.P., Rybachenko, A.I., and Kazakov, A.A., Fiziko-khimicheskie i analiticheskie kharakteristiki flavonoidnykh soedinenii (Physichemical and Analytical Characteristics of Flavonoids), Rostov-na-Donu: Rost. Gos. Univ., 1988.
Sauerwald, N., Schwenk, M., Polster, J., and Bengsch, E., Z. Naturforsch. A, 1998, vol. 53, p. 315.
Electronic Resource, http://www.boomer.org/pkin/PK01/PK2001237.html
Dubber, M.-J., PhD Thesis, Rhodes Univ., Greece, 2005; cited by http://eprints.ru.ac.za/294/01/MJ-Dubber-PhD.pdf
Herrero-Martinez, J.M., Sanmartin, M., Roses, M., Bosch, E., and Rafols, C., Electrophoresis, 2005, vol. 26, no. 10, p. 1886.
Woude, H., PhD Thesis, Wageningen Univ., The Netherlands, 2005; cited by http://library.wur.nl/wda/dis-sertations/dis3929.pdf
Herrero-Martinez, J.M., Repolles, C., Bosch, E., Roses, M., and Rafols, C., Talanta, 2008, vol. 74, p. 1008.
Tungjai, M., Poompimon, W., Loefchutinat, C., Kothan, S., Dechsupa, N., and Naukhetkorn, S., The Open Drug Delivery J., 2008, vol. 2, p. 10.
Battino, R., Rettich, T.R., and Tominaga, T., J. Phys. Chem. Ref. Data, 1983, vol. 12, no. 2, p. 163.
Vesilind, P.A. and Morgan, S.M., Introduction to Environmental Engineering, Boston: Brooks/Cole, 2003, 2nd ed.
Abraham, M.H., Whiting, G.S., Carr, P.W., and Onyang, H., J. Chem. Soc. Perkin Trans., 1998, no. 2, p. 1385.
Electronic Resources, http://www.thermidaire.on.ca/do.html and http://cee.citadel.edu
Shchukarev, S.A. and Tolmacheva, T.A., Zhurn. strukt. khim., 1968, vol. 9, no. 1, p. 21.
Golovanov, I.V. and Zhenodarova, S.M., Zhurn. obshch. khim., 2005, vol. 75, no. 11, p. 1879.
Leo, A., Hansch, C., and Eikins, D., Chem. Rev., 1971, vol. 71, no. 6, p. 525.
Zenkevich, I.G., Ishchenko, E.V., Makarov, V.G., Makarova, M.N., and Selezneva, A.I., Zh. Obshch. Khim., 2008, vol. 78, no. 9, p. 1449 [Russ. J. Gen. Chem. (Engl. Transl.), vol. 78, no. 9, p. 1682].
Nierenstein, M. and Whedale, M., Ber. Deut. Chem. Ges., 1912, vol. 44, p. 3487.
Matsuura, T., Matsushima, H., and Sakamoto, H., J. Am. Chem. Soc., 1967, vol. 89, p. 6370.
Matsuura, T., Matsushima, H., and Nakashima, R., Tetrahedron, 1970, vol. 26, p. 435.
Brown, S.B., Rajananda, V., Holroyd, J.A., and Evans, E.G.V., Biochem. J., 1982, vol. 205, p. 239.
Zenkevich, I.G., Eshchenko, A.Yu., Makarova, S.V., and Utsal’, V.A., Rastitel’lnye Resursy, 2007, vol. 43, no. 2, p. 111.
Zenkevich, I.G., Eshchenko, A.Yu., Makarova, S.V., Vitenberg, A.G., Dobryakov, Yu.G., and Utsal, V.A., Molecules, 2007, vol. 12, no. 3, p. 654.
Khaled, K.A. and El-Sayed, Y.M., J. Liquid Chromatogr. Rel. Technol, 2000, vol. 23, p. 455.
Rudakov, O.B., Vostrov, I.A., Fedorov, S.V., Filippov, A.A., Selemenev, V.F., and Pridantsev, A.A., Sputnik khromatografista. Metody zhidkostnoi khromatografii (Methods of Liquid Chromatography: A Handbook), Voronezh: Izd. “Vodolei”, 2004.
Razin, V.V. and Kostikov, R.R., Zadachi i uprazhneniya po organicheskoi khimii (Questions and Exercises on Organic Chemistry), St. Petersburg: St. Petersburg Gos. Univ., 2007.
Zborowski, K., Grybos, R., and Proniewich, L.M., J. Phys. Org. Chem., 2005, vol. 18, p. 250.
Demidovich, B.P., Lektsii po matematicheskoi teorii ustoichivosti (Lectures on Mathematical Stability Theory), Moscow: Nauka, 1967.
Author information
Authors and Affiliations
Additional information
Original Russian Text © I.G. Zenkevich, S.V. Guschina, 2010, published in Zhurnal Analiticheskoi Khimii, 2010, Vol. 65, No. 4, pp. 382–387.
Rights and permissions
About this article
Cite this article
Zenkevich, I.G., Guschina, S.V. Determination of dissociation constants of species oxidizable in aqueous solution by air oxygen on an example of quercetin. J Anal Chem 65, 371–375 (2010). https://doi.org/10.1134/S1061934810040064
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934810040064